
From Yellowstone Science 23(2): Pages 17-25.
One of the many benefits of studying grizzly bears in Yellowstone is they are a popular topic of conversation. The interest in Yellowstone grizzly bears is almost universal and for good reason, the Greater Yellowstone Ecosystem (GYE) invokes images of a wild, untamed landscape with grizzly bears as one of its most iconic species. In such conversations about bears, 9 out of 10 times the first question is "How many grizzly bears are there?" Many people expect a simple, straightforward answer. Although we usually have an answer, what's behind the answer is by no means simple. Like other elusive animals, grizzly bears are notoriously difficult to study. They occur in low densities, are active at times when observation is difficult (early morning and late evening), and use remote habitats and rough terrain. Only through a concerted, long-term research and monitoring effort have we begun to understand many fascinating aspects of grizzly bear population demographics (the statistical study of populations).
The Interagency Grizzly Bear Study Team (Study Team) started population studies in 1973 and have continued to-date, building one of the longest-running and most comprehensive biological datasets of any large carnivore in the world. To keep track of what the grizzly bear population is doing, the Study Team deploys a number of monitoring techniques ranging from observing bears in the field, to aerial observation and radio tracking from planes, to cutting-edge technologies, such as Global Positioning System (GPS) tracking with satellite transmitters. One of the primary jobs of the Study Team is to provide periodic assessments of the GYE grizzly bear population to assist managers with potential decision points in the management process. Managers often want to know the population size before deciding on appropriate actions. Additionally, monitoring information provided by the Study Team helps managers develop knowledge of how the population will likely respond to various types of environmental changes. In summary, one of our most important tasks is to count bears, which sounds easy, but doing so in practice and with scientific rigor is a very challenging task. Monitoring Grizzly Bears Early research efforts were dedicated to developing reliable population monitoring techniques that did not involve marking (e.g., numbered ear tags) or radio-collaring bears (figure 1), which was not permitted for several years after the Study Team was established. The first successful technique to be implemented involved counts of a specific segment of the population: females with cubs-of-the-year. This population segment was selected because females with cubs can be reliably distinguished from other bears (lone bears of either sex or females with yearling or two-year-old offspring), and they also represent the most crucial reproductive segment of the population. For some highly visible animals, such as elk (Cervus elaphus), a single aerial survey may provide a reliable estimate of an entire herd. However, bears are far less visible;therefore, sightings of females with cubs must be tallied over an entire spring and summer. Because observations occur over a long period, it was necessary to develop a rule set that assigns each observation to a particular "unique" female (Knight et al. 1995). Based on grizzly bear biology, the Study Team used information on the size of individuals' home ranges and size of family groups to assign an observation to either a previously observed or new female with cubs. For example, if a female with two cubs is observed only 5 kilometers from a previous observation of a female with two cubs, the observation is considered a resighting rather than a new unique female with cubs. It was recognized that these rules were not perfect;however, if errors occurred, the resulting count was conservative and was not designating multiple sightings as different females unless there was strong evidence. This reduced the risk of overestimating numbers of females with cubs and the overall population estimates derived from those counts, which was important for a population that did not show signs of growth until the mid-1980s. This method was adopted as part of the Grizzly Bear Recovery Plan in 1993. Further improvements to this technique occurred through the 1990s and early 2000s, which included a refinement to incorporate an estimate of females not observed, referred to as the "Chao2 estimator," named after the scientist who developed the technique (Keating et al. 2002, Cherry et al. 2007). Another enhancement was a statistical technique that would inform managers if a change in the trajectory of the population had occurred, essentially a "monitoring trigger" for the population (Harris et al. 2007). For example, a graph of a population growing at a constant rate each year will show a linear increase with each additional year of data.If this relationship changes from a linear, straight line to a nonlinear, curved line over time, it would signal a change in population trend. This monitoring trigger was implemented in 2007. In 2011 we detected that the growth rate of the Yellowstone grizzly bear population, which was linear during the 1980s and 1990s, had started to level off (figure 2). Further investigations showed this slowing of population growth likely started about a decade prior, around the early 2000s. For the Study Team, there was a very rewarding aspect to this result. Detecting a change in population growth meant the monitoring system set up by the Study Team had worked and could provide important information for managers. One might even call this a "textbook" example of what we teach students in wildlife management curricula throughout the country: set up a population monitoring system, establish triggers that indicate when a change has occurred, study the details of that change, and adjust management accordingly. Of course, you may wonder why we got excited about detecting this change a decade after it started? The reality is that monitoring wildlife populations, grizzly bears in particular, is fraught with challenges. One important reason for this is that the reproductive rates of grizzly bears are very low: grizzly bear generation time is about 10.5 to 13.5 years, which means it takes that long for a female grizzly bear to replace herself in the population. Detecting changes in a grizzly bear population simply takes time. Data from the three years after 2011 show higher estimates (figure 2), but because of annual variation we do not yet consider that indicative of a new period of population growth. Indeed, a preliminary estimate for 2015 reinforces the notion that the population remains stable. Changes in Vital Rates Since population growth had changed, we initiated a comprehensive demographic review in the fall of 2011 to investigate in detail what caused the slowing of population growth. Using studies by Schwartz et al. (2006) for the period 1983–2001 as a basis for comparison, we reevaluated "vital rates" of GYE grizzly bears for the period 2002–2011. Vital rates refer to demographic measures of the population, such as survival of different age classes and reproductive output. Vital rates can also be combined to estimate annual population growth, symbolized by lambda (see page 22). For the period 1983–2001, Schwartz et al. (2006) observed a robust rate of annual population growth of 4.2-7.6%. However, our analysis of the 2002–2011 data indicated this annual growth had slowed to about 0.3-2.2% (Interagency Grizzly Bear Study Team [IGBST] 2012). The proximate causes of this slower growth were lower survival rates among the cub and yearling age classes and a slight decline in fecundity (measured as the number of female cubs produced per breeding-age female per year;table 1). Survival of independent females (2 years or older) did not change between the two time periods, but survival of adult males actually increased from the earlier to the later time period. Recent analysis, based on life histories of more than 1,200 grizzly bears collected since the 1970s, suggests the recruitment of younger individuals has decreased as the proportion of older bears in the population has increased. Of course, the real question is "What do all these new findings tell us about the population biology of Yellowstone grizzly bears and the ultimate cause of these changes?" What are Potential Causes of Demographic Changes? Recent data suggest there is a spatial component to these changing demographics. Previous observations based on data from 1983 to 2001 (Schwartz et al. 2006) indicated that Yellowstone, the core area for grizzly bears in the GYE, had already shown signs of stabilizing numbers. It also indicated the most vigorous population growth occurred in the zone outside Yellowstone but within the Grizzly Bear Recovery Zone (a zone designated by the U.S. Fish and Wildlife Service to implement recovery; figure 3).Areas outside the recovery zone were essentially "sink" areas, where the mortality rate exceeded the birth rate. Findings from our analyses of 2002–2011 data showed the process of population stabilization had expanded to the entire Grizzly Bear Recovery Zone (IGBST 2012) and areas outside the zone functioned less as population sinks. Throughout both periods of robust and slower population growth, grizzly bears expanded their distribution in the GYE, recolonizing some areas where grizzly bears have been absent for over a 100 years ("Shorts: Grizzly Bears and Army Cutworm Moths," this issue). By 2010, grizzly bears occupied around 50,280 km2 (12.4 million acres) with few areas of prime habitat unoccupied (Bjornlie et al. 2014a). In other words, population growth rates have become more similar across the entire ecosystem as the population expanded, suggesting the population is reaching carrying capacity (a term used to define the long-term population size that can be sustained) given available space, food, and other resources in the environment. Case closed, right? No, not quite. Whereas the data indicated the grizzly population may be reaching carrying capacity in many portions of the ecosystem, managers wanted to know what the ultimate cause of this may have been. Was it the "natural" process of a population simply reaching high numbers (also referred to as density-dependence), or could carrying capacity have been substantially reduced as a consequence of recent changes in availability of food resources? The most notable change in a grizzly bear food resource occurred with whitebark pine trees (Pinus albicaulis), the seeds of which are a high-calorie fall food for grizzly bears preparing for hibernation. High mortality of whitebark pine trees has occurred throughout the GYE since the early 2000s, primarily due to a native forest insect, the mountain pine beetle (Dendroctonus ponderosae; see "How Important is Whitebark Pine to Grizzly Bears?," this issue), which could have lowered carrying capacity. If the latter was the case, managers may be more inclined to take a more cautious approach than if the population had naturally reached a stable level at a higher carrying capacity. We set out to study this question by comparing the association of whitebark pine decline and grizzly bear density with survival and reproduction rates based on three decades of data (1983–2012; van Manen et al. 2015). It is very difficult to tease apart potential effects of food versus bear density, so we focused on developing ways to measure the degree of whitebark pine's impact and grizzly bear density experienced by each individual bear in our analyses, both of which varied immensely throughout the GYE and over time. We detected no link between survival of independent bears and density or between their survival and whitebark pine decline.In fact, as we noted previously, survival of independent females (2 years or older) was high (95%) and has essentially not changed in 30 years. Survival of independent males increased during the decade of the 2000s from 87% to 95%.In contrast, survival of dependent bears (cubs and yearlings) declined and the drop in cub survival was most apparent among those bears that lived at high densities. There was no evidence of an association of cub and yearling survival with whitebark pine decline, but there was evidence for a link between reproduction and bear density. By concentrating our analysis on females with no offspring (i.e., those most likely to successfully breed), we found their probability of having cubs the next year declined slightly during the 1990s, prior to whitebark pine decline;and this decline was greater in areas with higher bear densities. Knowing that grizzly bear density may be a key factor, we next investigated potential drivers of how bear density may be affecting population growth. This is where things get really interesting. Potential Drivers of Density Dependence As a population approaches carrying capacity and factors start acting on the population as density increases, a predicted sequence of events for long-lived vertebrates is that juvenile mortality increases, females tend to initiate breeding later in life, reproductive output tends to decline, and, ultimately, adult mortality increases (Eberhardt 2002). We know from studies in Scandinavia and Alaska that cub survival is a potential density-dependent factor contributing to population regulation among bear populations (Swenson et al. 1997, 2001, Miller et al. 2003). Could it be that more older males in the population, because of their increased survival, are somehow related to lower survival of younger bears? Intraspecific killing by males may function as a direct density-dependent effect on cub survival, but there has been vigorous debate as to what the potential biological drivers may be. Studies in Scandinavia support the notion of sexually-selected infanticide (male bears kill cubs they did not sire to create new mating opportunities;Swenson et al. 1997, 2001);whereas, North American studies indicate a greater vulnerability of cubs to various sources of mortality, including intraspecific killings, for populations nearing carrying capacity (Miller et al. 2003). Regardless of this debate, males are primarily responsible for intraspecific killing, and we hypothesize there is a connection with the increased survival of independent-aged males (table 1) and increased mortality of cubs since the early 2000s. We have field observations of males killing cubs, including several instances where the mother was killed as well, presumably while defending her cubs (e.g., in Lamar Valley and Hayden Valley). The frequency of these events seems to have increased in the last 10 years compared with previous decades. Although we have some evidence supporting this hypothesis, we always investigate potential alternative explanations as well. Alternative Explanations The first alternative explanation may be "interference competition," which refers to constraints on feeding efficiency of subordinate individuals (often juveniles or females with cubs) because of the presence of dominant individuals. Such competition can limit access to foods for subordinates, even when foods are plentiful. If conspecifics are potentially predatory, as in grizzly bears, increased vigilance may intensify this effect at higher population densities.Studies in Alaska (Nevin and Gilbert 2005) and Scandinavia (Steyaert et al. 2013) have demonstrated reduced energy intake among females with cubs as a consequence of avoidance of more dominant bears, especially large males. However, similar evidence of interference competition has not been observed in the GYE so far. Therefore, we cannot conclude whether or not it was a contributing factor to the slowing of population growth. Another alternative explanation of the decline in cub and yearling survival may be the reintroduction of gray wolves (Canis lupus) into Yellowstone National Park in 1995–1997. However, only four incidents of wolf predation on cubs or yearlings have been documented since 2001 (Gunther and Smith 2004, IGBST, unpublished data) so support for this explanation is not very strong. Conclusions Multiple lines of evidence (IGBST 2013) support our hypothesis that the slowing of population growth of Yellowstone grizzly bears is associated with increasing density of grizzly bears rather than the decline of a calorie-rich fall food resource, whitebark pine (van Manen et al. 2015). Nevertheless, population changes brought about by density effects may still be linked with food resources and carrying capacity of the environment. We considered the possibility that decline of whitebark pine and other resources (see "Response of Grizzly Bears to Changing Food Resources in the GYE," this issue) reduced carrying capacity, which could have reduced cub survival and reproductive transitions in a density-dependent fashion through direct competition for resources. This effect would be difficult to separate from that of interference competition as discussed previously. If this were the case, we would have expected home-range size and movements to increase (McLoughlin et al. 2000), bears to have relied on lower-energy food resources (McLellan 2011), and body condition to have declined as a consequence (Robbins et al. 2004). To date, there is little evidence for these conditions in the GYE;female home ranges have decreased in size where bear densities have increased (Bjornlie et al. 2014b), daily movements have not changed for females or males during fall hyperphagia (i.e., increased appetite before hibernation;Costello et al. 2014), bears continue to use high-quality foods (Fortin et al. 2013), body mass has not declined (Schwartz et al. 2014), and percent body fat among females in fall has not changed (IGBST 2013). Finally, current evidence suggests grizzly bears responded to declines in whitebark pine (Costello et al. 2014) and cutthroat trout (Oncorhynchus clarkii; Fortin et al. 2013) by shifting their diets, indicating this opportunistic omnivore was able to compensate for the loss of these particular foods. Literature Cited Bjornlie, D.D., D.J. Thompson, M.A. Haroldson, C.C. Schwartz, K.A. Gunther, S.L. Cain, D.B. Tyers, K.L. Frey, and B. Aber. 2014a. Methods to estimate distribution and range extent of grizzly bears in the Greater Yellowstone Ecosystem. Wildlife Society Bulletin 38:182–187. Bjornlie, D.D., F.T. van Manen, M.R. Ebinger, M.A. Haroldson, D.J. Thompson, and C.M. Costello. 2014b. Whitebark pine, population density, and home-range size of grizzly bears in the Greater Yellowstone Ecosystem. PLoS ONE 9:e88160. Cherry, S., G.C. White, K.A. Keating, M.A. Haroldson, and C.C. Schwartz. 2007. Evaluating estimators of the numbers of females with cubs-of-the-year in the Yellowstone grizzly bear population. Journal of Agricultural, Biological, and Environmental Statistics 12:195–215. Colinvaux, P.A. 1986. Ecology. Wiley &Sons, New York, New York, USA. Costello, C.M., F.T. van Manen, M.A. Haroldson, M.R. Ebinger, S. Cain, K. Gunther, and D.D. Bjornlie. 2014. Influence of whitebark pine decline on fall habitat use and movements of grizzly bears in the Greater Yellowstone Ecosystem. Ecology and Evolution 4:2004–2018. Eberhardt, L.L. 2002. A paradigm for population analysis of long-lived vertebrates. Ecology 83:2841–2854. Fortin, J.K., C.C. Schwartz, K.A. Gunther, J.E. Teisberg, M.A. Haroldson, and C.T. Robbins. 2013. Dietary adaptability of grizzly bears and American black bears in Yellowstone National Park. Journal of Wildlife Management 77:270–281. Gunther, K.A., and D.W. Smith. 2004. Interactions between wolves and female grizzly bears with cubs in Yellowstone National Park. Ursus 15:232–238. Haroldson, M.A., K.A. Gunther, D.P. Reinhart, S.R. Podruzny, C. Cegelski, L. Waits, T. Wyman, and J. Smith. 2005. Changing numbers of spawning cutthroat trout in tributary streams of Yellowstone Lake and estimates of grizzly bear visiting streams from DNA. Ursus 16:167–180. Haroldson, M.A., C.C. Schwartz, K.C. Kendall, K. Frey, K.A. Gunther, D. Moody, and D. Paetkau. 2010. Genetic analysis of individual origins supports isolation of grizzly bears in the Greater Yellowstone Ecosystem. Ursus 21:1–13. Harris, R.B., G.C. White, C.C. Schwartz, and M.A. Haroldson. 2007. Population growth of Yellowstone grizzlies: uncertainty, correlation, and future monitoring. Ursus 18:168–178. Interagency Grizzly Bear Study Team. 2012. Updating and evaluating approaches to estimate population size and sustainable mortality limits for grizzly bears in the Greater Yellowstone Ecosystem. Interagency Grizzly Bear Study Team, U.S. Geological Survey, Northern Rocky Mountain Science Center, Bozeman, Montana, USA. Interagency Grizzly Bear Study Team. 2013. Response of Yellowstone grizzly bears to changes in food resources: A synthesis.Report to the Interagency Grizzly Bear Committee and Yellowstone Ecosystem Subcommittee. Interagency Grizzly Bear Study Team, U.S. Geological Survey, Northern Rocky Mountain Science Center, Bozeman, Montana, USA. Keating, K.A., C.C. Schwartz, M.A. Haroldson, and D. Moody. 2002. Estimating numbers of females with cubs-of-the-year in the Yellowstone grizzly bear population. Ursus 13:161–174. Knight, R.R., B.M. Blanchard, and L.L. Eberhardt. 1995. Appraising status of the Yellowstone grizzly bear population by counting females with cubs-of-the year. Wildlife Society Bulletin 23:245–248. Krebs, C.J. 2009. Ecology: the experimental analysis of distribution and abundance. Benjamin Cummings. San Francisco, California, USA. McLellan, B.N. 2011. Implications of a high-energy and low-protein diet on the body composition, fitness, and competitive abilities of black (Ursus americanus) and grizzly (Ursus arctos) bears. Canadian Journal of Zoology 89:546–558. McLoughlin, P.D., S.H. Ferguson, and F. Messier. 2000. Intraspecific variation in home range overlap with habitat quality: a comparison among brown bear populations. Evolutionary Ecology 14:39–60. Miller, S.D., R.A. Sellers, and J.A. Keay. 2003. Effects of hunting on brown bear cub survival and litter size in Alaska. Ursus 14:130–152. Nevin, O.T., and B.K. Gilbert.2005. Measuring the cost of risk avoidance in brown bears: further evidence of positive impacts of ecotourism. Biological Conservation 123: 453–460. Robbins, C.T., C.C. Schwartz, and L.A. Felicetti. 2004. Nutritional ecology of ursids: a review of newer methods and management implications. Ursus 15:161–171. Schwartz, C.C., J.K. Fortin, J.E. Teisberg, M.A. Haroldson, C. Servheen, C.T. Robbins, and F.T. van Manen. 2014. Body and diet composition of sympatric black and grizzly bears in the Greater Yellowstone Ecosystem. Journal of Wildlife Management 78:68–78. Schwartz, C.C., M.A. Haroldson, G.C. White, R.B. Harris, S. Cherry, K.A. Keating, D. Moody, and C. Servheen. 2006. Temporal, spatial, and environmental influences on the demographics of the Yellowstone grizzly bear. Wildlife Monographs 161: 1-8. Schwartz, C.C., S.D. Miller, and M.A. Haroldson. 2003. Grizzly bear. Pages 556–586 in G.A. Feldhamer, B.C. Thompson, and J.A. Chapman, editors. Wild mammals of North America: Biology, management, and conservation. Second edition.The Johns Hopkins University Press, Baltimore, Maryland, USA. Steyaert, S.M.J.G., C. Reusch, S. Brunberg, J.E. Swenson, K. Hackländer, and A. Zedrosser. 2013. Infanticide as a male reproductive strategy has a nutritive risk effect in brown bears. Biology Letters 9:20130624. Swenson, J.E., F. Sandegren, S. Brunberg, and P. Segerström. 2001. Factors associated with loss of brown bear cubs in Sweden. Ursus 12:69–80. Swenson, J.E., F. Sandegren, A. Söderberg, A. Bjärvall, R. Franzén, and P. Wabakken. 1997. Infanticide caused by hunting of male bears. Nature 386:450–451. Taylor, M.K., and D.L. Garshelis. 1994. Density-dependent population regulation in black, brown, and polar bears. International Conference on Bear Research and Management. Monograph Series No. 3. Teisberg, J.E., M.A. Haroldson, C.C. Schwartz, K.A. Gunther, J.K. Fortin, and C.T. Robbins. 2014. Contrasting past and current numbers of bears visiting Yellowstone cutthroat trout streams. Journal of Wildlife Management 78:369–378. van Manen, F.T., M.A. Haroldson, D.D. Bjornlie, M.R. Ebinger, D.J. Thompson, C.M. Costello, and G.C. White. 2015. Density dependence, whitebark pine, and vital rates of grizzly bears. Journal of Wildlife Management, in press. 
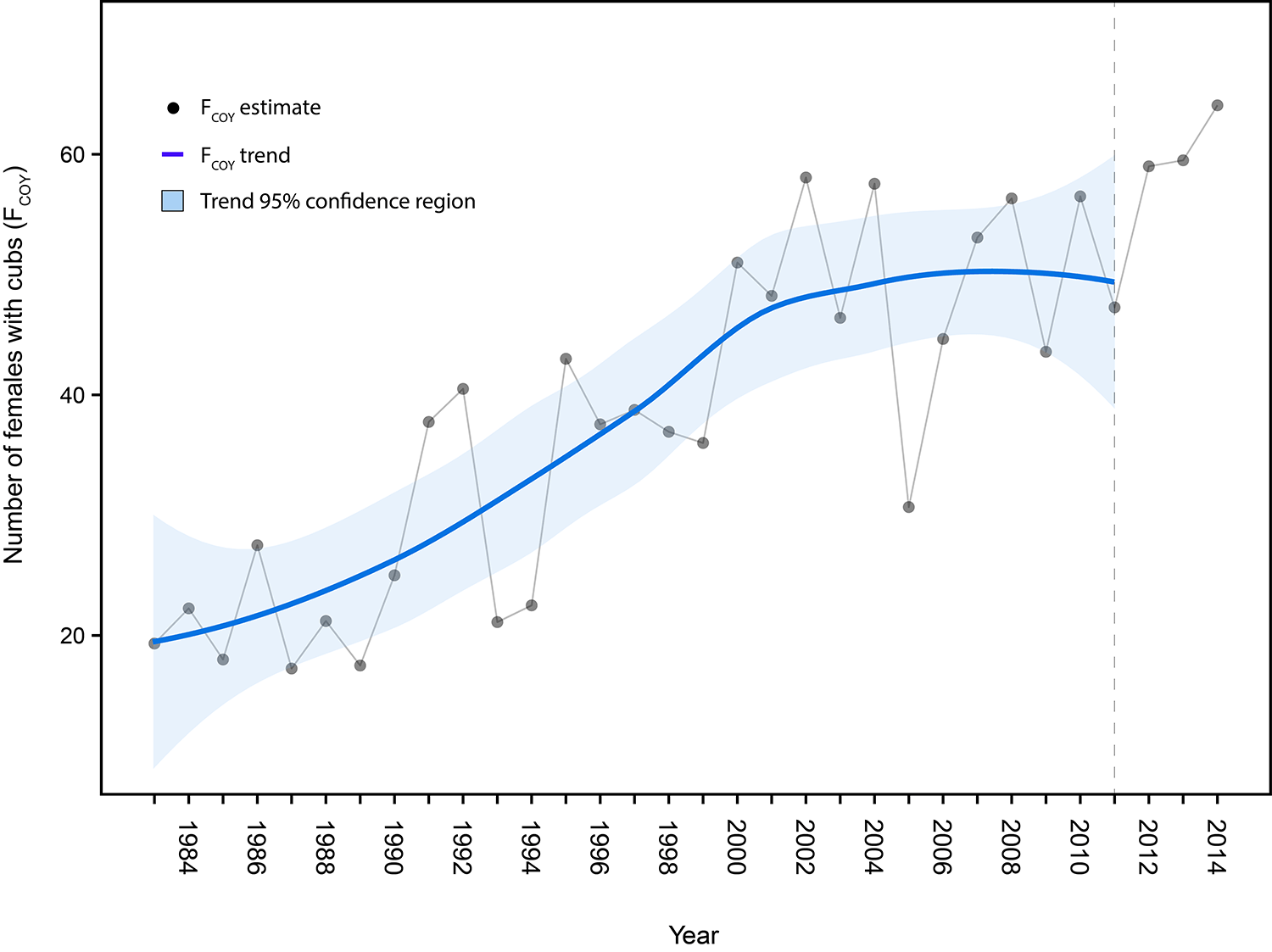
Figure 2. Estimated number (Chao2 estimator;Keating et al. 2002) and population trend (based on locally weighted scatter-plot smoothing) of unique females with cubs of the year (FCOY), Greater Yellowstone Ecosystem, 1983-2014. 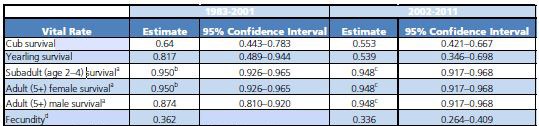
a Animals with unresolved fates were censored for analysis at last contact 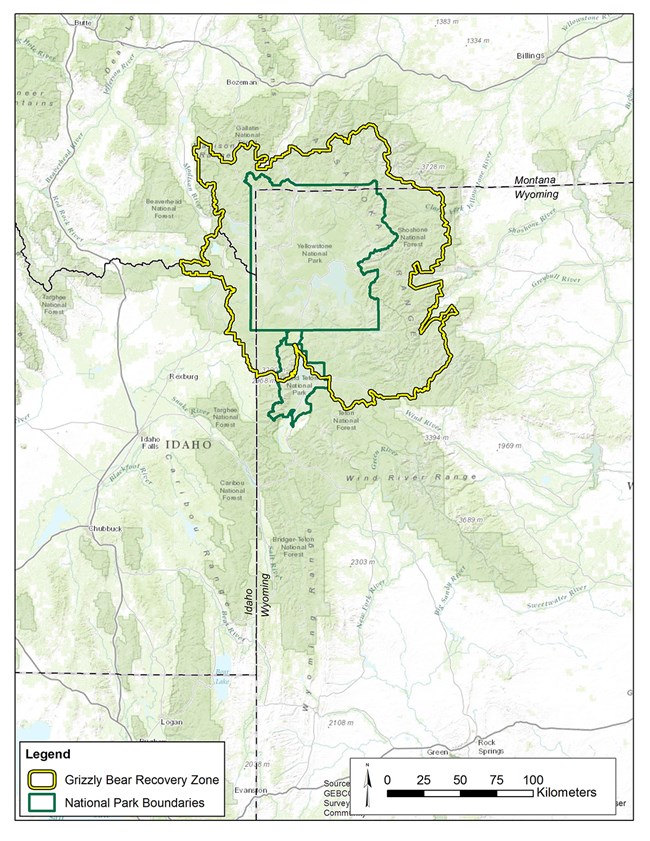
|
Last updated: December 21, 2015
