|
Winter 2022 Volume 22 Issue 2

Top photo: USFS, Middle photo: George Gruell, Bottom photo: NPS Shifting BaselinesBy Bryan Hamilton, Integrated Resource Program ManagerWait!! What? This wasn’t always a forest? 
The Nature Conservancy. Illustrator: Kelly Finan, Adapted from: Threat-based land Management: A Field Guide (The SageShare Partnership) Soil formation is not only a geologic process, but a direct reflection of the plants that formed the soils over thousands of years. Based on soil composition, we know that in some forested areas, trees that are present today were not a significant contributor to the creation of those soils. Instead, soil was formed by sagebrush and grasses, not the acidic needles of a conifer forest. Dendrochronology provides more strong evidence. Pinon and juniper trees can live extraordinarily long lives. The oldest recorded Utah juniper lived nearly 2,000 years; the oldest single needle pinon pine 900 years (Weisberg and Ko, 2012). If the increase in trees is due to fire exclusion, we would expect relatively young trees in former sagebrush habitat. Indeed, almost all trees are less than 100 years old, aligning well with the beginning of colonization and the fire exclusion era. Ancient pinons and junipers are often found in pockets within or adjacent to sagebrush habitat, but these stands are scattered and limited to rocky slopes with thin soils, areas protected from fires. Old growth trees on these sites show regular fire scars, further documentation of the frequency of low intensity fires of the past. Historic documents and photographs offer still more evidence. Journal entries, place names, wildlife observations, and livestock stocking records indicate a more open, relatively treeless Great Basin as compared to today. Historic photos further corroborate the increase in pinon and juniper and subsequent decrease in sagebrush and grasslands. Over time, we have forgotten about “good fire” and the historic, open sagebrush landscapes it maintained. We’ve come to view widespread pinyon juniper forests as a permanent part of our environment. But these young forests have major effects on plants and animals. Wildlife, like sage grouse, pygmy rabbits, and yellow-bellied marmots need open sagebrush habitat to survive. Other species like mule deer and migratory birds have declined due to conifer encroachment. Less water is available in streams and springs and there is less native grass and more barren soil. The land is less productive and less diverse. Fire seemed like the obvious solution to restore sagebrush. But fire itself now presents a challenge. In pinyon juniper forests, dense flammable fuels create catastrophic fires that are hard to control and can damage soils. Following these fires, cheatgrass, an invasive annual, can take over. Cheatgrass is now found on over 17 million acres in the Great Basin and has a negative effect on biodiversity and ecosystem services. Rather than using fire, chainsaws or masticators are used to remove conifers. Sagebrush and other native species, like bluebunch wheatgrass or penstemon, are often seeded on restoration sites after conifer removal. Our success in restoring sagebrush varies but is often better in places with deeper soils, native plants in the understory, and higher precipitation. In addition to restoring sagebrush, conifer removal treatments are designed to recover quickly after wildfires and to resist invasion by cheatgrass. Maintaining sagebrush may require regular maintenance to remove young conifers before they can establish. Restoration projects are carefully monitored to ensure project goals are met. The sagebrush ocean is the Great Basin. But this vast, beautiful landscape is threatened. Fire exclusion and conifer encroachment have slowly changed sagebrush habitats, leaving these areas vulnerable to catastrophic wildfires and invasion by cheatgrass. As our baselines shift, we may come to see pinyon juniper forests as a fixture, rather than seeing the sagebrush habitat that the conifers have replaced. But active restoration through conifer removal can restore the sagebrush ocean and the wildlife that depend on it. References: Chambers, J. C. 2008. Fire and the Great Basin, p. 33-37. In: Collaborative management and research in the Great Basin - examining the issues and developing a framework for action. J. C. D. Chambers, Nora; Evenden, Angela (ed.). Gen. Tech. Rep. RMRS-GTR-204. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. Knick, S. T., A. L. Holmes, and R. Miller. 2005. The role of fire in structuring sagebrush habitats and bird communities. Studies In Avian Biology. 30:1-13. Miller, R. F., and R. J. Tausch. 2001. The role of fire in pinyon and juniper woodlands: a descriptive analysis. Proceedings of the Invasive Species Workshop: the Role of Fire in the Control and Spread of Invasive Species. Fire Conference 2000: the First National Congress on Fire Ecology, Prevention, and Management. Miscellaneous Publication No. 11, Tall Timbers Research Station, Tallahassee, FL.:15-30. Soga, M., and K. J. Gaston. 2018. Shifting baseline syndrome: causes, consequences, and implications. Frontiers in Ecology and the Environment. 16:222-230. Weisberg, P. J., and D. W. Ko. 2012. Old tree morphology in singleleaf pinyon pine (Pinus monophylla). Forest Ecology and Management. 263:67-73. Welch, B. L. 2005. Big sagebrush: A sea fragmented into lakes, ponds, and puddles. Gen. Tech Rep. RMRS-GTR-144. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station:210. Back to Top Back to The Midden main page 
Brianna Patterson Hidden Stories of Snake Creek CaveBy Brianna Patterson, Archaeologist, Great Basin InstituteFor thousands of years, humans have entered caves and made their mark. From rock writing to historic inscriptions, these cultural resources capture a moment of the composer’s life. Such is the case for the more than 200 historic inscriptions within Snake Creek Cave in Great Basin National Park. Archaeologists see these entries as more than just names and dates. They use the information left behind to piece together past lives. It’s the stories archaeologists tell that are at the heart of our work.A small crew composed of volunteers and one archaeologist recently started uncovering the history of Snake Creek Cave’s former explorers. Within three hours, the crew had recorded 104 inscriptions, which cover a span of time from 1885 to 1978. These inscriptions include several fascinating entries such as “Kenneth B. Vanda H. 1932 Sweethearts”, “Francis April 21, 1935 3rd Grade”, and a potential cattle brand symbol made using carbide. 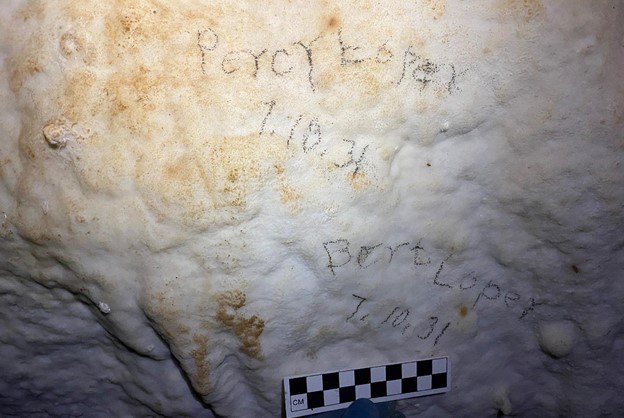
Brianna Patterson. Though there is still much work left, the crew’s efforts have already made a connection to a man named Bert Loper. Through genealogical research, the crew discovered Bert was born in 1913 in Burbank, Utah a little over twenty miles from the site. In 1930, he first entered the cave at seventeen-years-old and returned the next year with his thirteen-year-old brother, Percy Loper. In 1935, Bert moved to Las Vegas, Nevada to work as a steel cleaner and painter at the Hoover Dam. Sadly, Bert died on November 17th after roughly three months on the job. He was electrocuted while working on the rails of a crane in the power house and fell sixty feet. 
Wade Plafcan Life at the Top: Range Shifts of Four High Elevation PlantsBy Wade Plafcan and Thomas Albright, Researchers, University of Nevada, RenoStarting in fall of 2021, Great Basin National Park, the University of Nevada, Reno, and botanist Jan Nachlinger began a project to track changes in recent and future distributions of four rare alpine plant species. 
Wade Plafcan With GPS units, sturdy boots, and lots of water, our team surveyed thousands of acres for plant locations in the summers of 2021 and 2022. On top of this, we were lucky that some forward-looking naturalists had conducted extensive surveys in 2004-2006. This gave us the opportunity to look for changes: places where the plants may be dying off and locations where new populations may be establishing. Overall, surveyors found about half of the plants occupying the same locations from the previous survey. This is an impressive survey effort, as finding a single plant sometimes takes 3-4 people on their hands and knees for several minutes! A comparative analysis between the two surveys revealed a potentially alarming trend. For all species, individuals found lower in the species’ elevational range were more likely to be absent, while the new and persistent populations occurred higher in elevation. This suggests an upward elevational range shift. 
But what about conserving these species into the future? The best way to ensure the preservation of species vulnerable to human-driven climate change is to reduce the emissions of greenhouse gasses globally. Beyond those measures, an effective conservation plan for rare species needs to identify locations of suitable habitat to prioritize and target conservation efforts. Species distribution models (SDMs) give insight into these locations and allow for prediction of current and future habitat. SDMs relate a species’ geographic location with environmental variables to produce predictive distribution maps. Plant locations were matched with climate, topography, and soil variables. Through this process we were able to quantify which environmental variables were important for each species. Current habitat maps are useful for finding new populations and as an estimate of total current habitat. 
NPS Photo/Joey Danielson Genetic Analysis of West Desert Bonneville Cutthroat TroutBy Joseph Danielson, Biological Science TechnicianGreat Basin National Park (GRBA) is currently working in collaboration with Brigham Young University (BYU) to determine genetic variation and purity among Bonneville Cutthroat Trout (BCT) populations in Nevada.The BCT is a subspecies of cutthroat trout that lived throughout the Bonneville Basin, which included Lake Bonneville (now the Great Salt Lake) and its tributaries, at the end of the last ice age. Once thought to be extinct, genetically pure populations have been found in recent decades in Utah, Idaho, Wyoming, and Nevada. The BCT is the only species of trout native to GRBA and east central Nevada. Careful management has allowed the BCT to remain off the endangered species list, but human encroachment, the introduction of non-native trout, an increase in fire severity, and worsening drought still threatens the species, particularly at the periphery of its range. The information gained through genetic analysis will be used to determine the source population(s) for the Baker Lake BCT introduction and for other future reintroductions and management actions. Current BCT populations in the region are isolated and rather small in numbers, particularly with the recent drought years, which can negatively affect the genetic diversity of these populations through inbreeding depression and genetic drift. Identifying these genetically diminished populations, and whether certain streams could benefit from inter-population translocations will help us maintain healthy populations of fish. 
NPS photo/Joey Danielson BYU’s analysis will shed light on how much genetic variation there is in fish within and among Nevada’s BCT streams and show if there has been any introgression with non-native trout species. Although historically BCT were the only trout species native to east central Nevada, the haphazard introduction of other trout species following white European settlement has led to BCT being either outcompeted and/or hybridized in many areas. Locally, the Rainbow Trout is of particular concern as it can successfully spawn with BCT to produce fertile hybrid offspring and has done so in GRBA streams in the past. Nevada’s populations of BCT were reestablished using few local source populations and we expect that the genetic analysis will reflect these different lineages. While these populations at the time were not considered to be bottlenecked, the size of the populations suggest that genetic diversity could be an issue in the future. Once the genetic analysis is complete, the Park will have information needed to proceed with introducing BCT into Baker Lake in 2023 as well as make decisions on how to optimally manage all the isolated BCT populations in Nevada. 
Jean Krejca Mark and Recapture Study for the Model Cave Harvestman, Sclerobunus ungulatusBy Shiloh McCollum, Biologist, Great Basin InstituteArachnids, particularly shy cavernicolous ones, keep their secrets close to their scute. In fact, they don’t like to spill the beans at all. In order to engage them in disclosing all their secrets we cannot whisper sweet nothings, instead hard science is called to the field of battle.Harvestmen are solitary, photophobic omnivores that can sometimes exceed, in biomass, the number of spiders1. Cavernicolous species often have less seasonal variation than surface species1, but overall, not a lot is known about them. Sometimes they have continuous reproduction, sometimes not. Sometimes they guard their eggs and juveniles, sometimes not. Sometimes they are highly vagile (like to move), sometimes not.1 Sclerobunus ungulatus, the Model Cave Harvestman, is endemic to Great Basin Caves and even less is known of their secrets. We do not know if they feed on feces or springtails1. We do not know much about their biology, population dynamics, vagility, or really any of their secrets. We do know that the type specimen was collected in 1952 from Model Cave2. It differs from the closely related Sclerobunus cavicolens by a variety of features, including a lack of pigmentation2. It also differs from Sclerobunus madhousensis by a longer second leg and a limited range (S. madhousensis is endemic to caves near Provo, UT)2. Its current range covers the Baker Creek Watershed and 2 high elevation caves3,4. Thus, we are introducing a mark and recapture study in order to gain more knowledge and plunder secrets like mad pirates… or mad scientists (really, a very fine line sometimes). 
Jean Krejca The study is conducted every month by the same two researchers to reduce variables. Each researcher searches a transect for 15 minutes, collects all the specimens of interest they find, and makes observations of other species nearby (possible prey items). Then the other researcher does the same for another 15-minute pass. Once captured, the specimens are marked with fluorescent, acrylic paint with unique identifiers. A different color is used for each capture date, and a different leg is painted based on the transect. 
Shiloh McCollum After a year of our study, data will be compiled and crunched using appropriate statistical methods1 to learn as much as we can about population size, size classes, life span, reproduction, and any other biological observations we can glean during our study. Using Model Cave, we can then begin to unravel S. ungulatus’ secrets.

Austin Koontz A Closer Look at Nevada PrimroseBy Austin Koontz, ResearcherAt the summit of Mt. Washington grows a rare plant with a unique backstory. Primula cusickiana var. nevadensis, the Nevada Primrose, is a perennial plant with showy purple petals (Panel D in image to the right), and was first described as its own species in 1967 by Noel Holmgren. It is part of the Primula cusickiana species complex: a group of related plants with similar morphologies found throughout the Great Basin. The other varieties in the complex are:
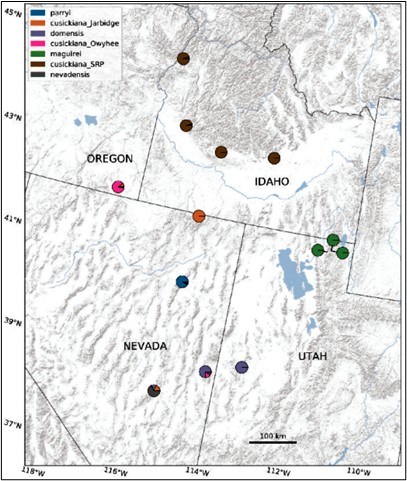
At the end of the Pleistocene 11,700 years ago, the Great Basin was a cool, relatively wet area featuring large lakes, such as the ancient Lake Lahontan and Lake Bonneville. At this time, the ancestor of Primula cusickiana was likely prevalent throughout the region. Over the course of the next 10,000 years (the Holocene), the Great Basin became hotter and drier. Mountaintops, however, continued to provide the cool conditions many organisms were used to; as the climate changed, these organisms retreated to these mountainous regions, which scientists call refugia. Primula was one of these organisms: as it retreated towards higher elevations to follow the cool, moist soils it prefers, its populations became fragmented. This fragmentation led to populations becoming more and more genetically unique, leading to the strong genetic divisions we see between populations today. Our research also shined a light on the unique nevadensis population on Mt. Washington. Our research suggests that this population of nevadensis is a hybrid between the domensis population (to the east in the House Range) and another nevadensis population further south (in the Grant Range). More needs to be done to characterize these two populations, but our work suggests that the Mt. Washington population (and indeed all populations) is very unique! I hope this work is built upon and can lead to the protection of these rare and unique plants! Interested readers can find out more about this work in the article here, published in Systematic Botany. 
NPS Photo Preserving Historic Lehman Orchardby Meg Horner, BiologistLehman Orchard changed for the better this past summer. With funding from the Southern Nevada Public Lands Management Act (SNPLMA) Conservation Initiative, park staff replaced the irrigation system, planted new fruit trees, installed new fencing, and designed and installed informational signs and a viewing platform. The upgrades and orchard restoration work completed this year will help preserve Lehman Orchard for years to come.The history of Lehman Orchard is lengthy and not well documented. The orchard was likely planted in the 1890s soon after Absalom S. Lehman settled near Lehman Caves and started promoting the caves as a tourist attraction. Records indicate Lehman planted fruit trees and constructed an aqueduct that brought water from Lehman Creek and nearby springs to irrigate the orchard. Four kinds of fruit trees were planted in the original orchard including apricot, apple, pear, and peach. Fruit from the orchard became an important source of food for the local community and income for Absalom Lehman and his family. Remarkably, a few of the apricots planted over a century ago are believed to be the same apricots still growing in the orchard today. 
NPS photo The most recent orchard restoration and preservation efforts began in 2017 with funding from the SNPLMA program as part of the Protection, Stabilization, and Restoration of the Lehman Caves Historic Area Project. Park staff researched the history of Lehman Orchard as well as the species and varieties of fruit trees growing in the local area. A greenhouse was constructed to grow seedlings for future planting. Seeds and cuttings were collected from the historic apricots in Lehman Orchard; Marianna root stock was purchased and grafted with ‘volunteer’ apples that were discovered several years earlier growing at two historic camp sites along Baker and Lehman Creeks; and Elberta variety heirloom peaches were purchased. Pear cuttings were collected from a local ranch, but seedlings did not survive. 
NPS Photo/Meg Horner Initial results from genetic testing solved a couple mysteries about the heirloom varieties of Lehman Orchard’s historic trees. One of the historic apricot samples was a match for Moorpark, a very old and highly regarded apricot variety dating back to the 1600s when the trees were brought from China to England for cultivation. Several of the other historic apricots in Lehman Orchard were likely seedlings from a Moorpark variety. There were no matches for the apples that were used for grafting from Baker or Lehman Creek. Further testing is planned to identify additional varieties and help preserve the historic and genetic character of Lehman Orchard. The NPS is committed to the preservation of Lehman Orchard, the legacy of the century-old apricots that still produce fruit every few years and sharing the story of this unique resource for another 100 years. Selected Publications about the Park
Upcoming Events

Illustration by Emily Hale More informationThe Midden is the Resource Management newsletter for Great Basin National Park. A summer and winter issue are posted each year. We welcome submissions of articles or drawings relating to natural and cultural resource management and research in the park. They can be sent to: Editor, Resource Management, Great Basin National Park, Baker, NV 89311 Or call us at: (775) 234-7331.Superintendent: James Woolsey Integrated Resource Program Manager: Bryan Hamilton Editor & Layout: Gretchen Baker What’s a midden?A midden is a fancy name for a pile of trash, often left by pack rats. Pack rats leave middens near their nests, which may be continuously occupied for hundreds, or even thousands, of years. Each layer of trash contains twigs, seeds, animal bones and other material, which is cemented together by urine. Over time, the midden becomes a treasure trove of information for plant ecologists, climate change scientists, and others who want to learn about past climatic conditions and vegetation patterns dating back as far as 25,000 years. Great Basin National Park contains many middens. |
Last updated: November 9, 2022
