Last updated: December 2, 2021
Article
Monitoring a Changing Bay
By Deanna Ochs
Is the fresh water pouring from rapidly metling glaciers changing the seawater in Glacier Bay? If so, what will that mean for marine life...and those who depend on it?
Phase II: 2015 to 2018
Is the fresh water pouring from rapidly metling glaciers changing the seawater in Glacier Bay? If so, what will that mean for marine life...and those who depend on it?
Project Dates
Phase I: 2011 to 2014Phase II: 2015 to 2018

NPS
Melting into Glacier Bay
"Glacier Bay waters may be experiencing ocean acidification on steroids." Lewis Sharman, Ecologist, Glacier Bay National Park and Preserve

NPS/ D. Ochs
Russ is one of several researchers analyzing the conditions for life in the seawater lapping at the steep fjord walls of Glacier Bay. Oceans the world over are becoming more acidic as they absorb increasing amounts of anthropogenic CO2 from the earth’s atmosphere. While levels vary from place to place, since the Industrial Revolution, average pH levels in ocean surface waters have decreased by 0.11. Because pH is measured on a logarithmic scale, this means the ocean’s acidity has increased by 30%. Despite this, at a pH of 8.1 seawater is still alkaline, or basic, and thus retains somewhat of a buffer against acidification. (Solutions with pH levels less than 7 are considered acidic). According to Park Ecologist Lewis Sharman, however, Glacier Bay waters may be experiencing ocean acidification on steroids.
Glacier Bay is in the midst of a period of rapid deglaciation. As the glaciers melt, most of this newly liberated glacial water is dumped directly into the bay. Fresh water is typically neutral or even slightly acidic. The greater the volume of fresh water that enters the bay, the more diluted the bay water becomes, and the lower its natural buffering capacity. This process can drive the pH lower.
What are the Impacts?

NOAA
How is the Park Studying the Issue?
NPS
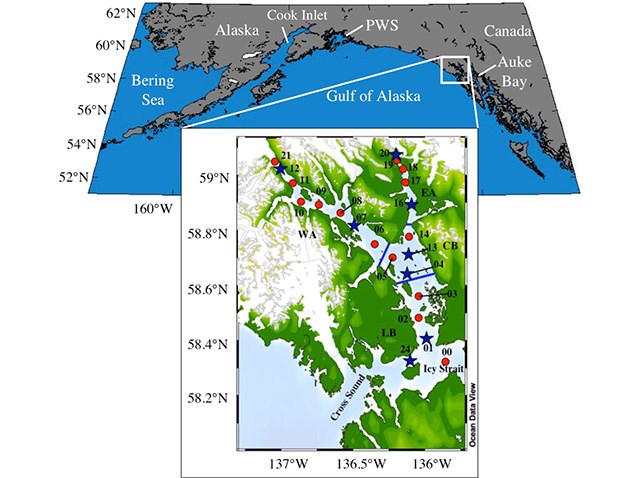
To conduct the study, researchers head out into the bay four to six times a year to collect seawater and plankton samples from 22 permanent stations. Most stations are located along the east and west arms of the bay, with a heavier concentration near the glaciers, where meltwater levels are highest. Two sites are located at the mouth of the bay to measure conditions of the water entering from the open sea.

NPS/D. Ochs

NPS/D. Ochs
While the study has not yet concluded, preliminary findings indicate that, indeed, Glacier Bay waters are becoming more acidic. It is also apparent that this change is more pronounced in summer and fall, when glaciers are melting fastest, and larger quantities of fresh water are entering the bay.
What this means for Russ’s prized zooplankton and the rest of Glacier Bay’s living organisms should become much clearer in the coming months. Results from Phase I of the study are available at https://www.nps.gov/articles/oceanacidificationinglacierbay.htm or at https://www.nps.gov/articles/aps-15-1-9.htm.
