Last updated: February 15, 2026
Article
Changes over 30 years in populations of the leafcutter ant Atta mexicana at Organ Pipe Cactus National Monument: An analysis of long-term environmental impacts on population size and survival

Photo courtesy of Alex C. Mintzer
Atta mexicana (F. Smith) at Organ Pipe Cactus National Monument, Arizona, provides an amazing example of survival and success at the northern limit of its range in the Sonoran Desert. Known as leafcutter ants, Atta spp. are ubiquitous objects of fascination in the dense forests of tropical America from Mexico to Argentina (Hölldobler and Wilson 1990), but their existence in the much drier environment of the Sonoran Desert of Arizona is a surprise to most observers. The main range of this ant species is in Mexico, with a southern limit in Guatemala and El Salvador (Smith 1963). These ants have been noted in an impressive diversity of habitats in Mexico, including oak and pine woodlands, mesquite grasslands, deserts, arid thorn scrublands, tropical short-tree deciduous forests, and tropical tall-tree deciduous and semideciduous forests (Smith 1963; Mintzer, personal observation based on extensive travel).
At Organ Pipe, this ant offers a unique interpretive opportunity, a natural history that has been related to thousands of visitors since 1987. Like other Atta species associated with tropical ecosystems, A. mexicana produces large central nests in this park, occupied by colonies that probably number in the millions of individual ants (fig. 1). These nests and colonies dwarf those produced by all other resident ant species (Mintzer 1987). The individual ants are large, and leaf-cutting and item-carrying behaviors of foraging ants draw attention from park visitors in the cooler months, when they are active during the daytime. At Organ Pipe, the colonies are dependable targets for interpretive walks led by ranger-naturalists (fig. 2).

Photo courtesy of Alex C. Mintzer
The first record of A. mexicana at Organ Pipe was provided by Byars (1949), who collected them near a gravel pit 1 km (0.6 mi) north of the current park headquarters. Another collection was made by entomologist R. E. Gregg and his party in 1948. W. S. Creighton visited Organ Pipe in 1952, but did not encounter them in the park; this species is not listed in his comprehensive monograph of North American ants (Creighton 1950). George and Jeanette Wheeler (1983) published a photograph of the superstructure of a nest along the Puerto Blanco Drive. National Park Service staff has been aware of these ants in the national monument since the 1970s. Staff encountered one colony along the west side of the vehicle maintenance yard in the mid-1970s, and found a second in Lost Cabin basin (fig. 3).
I first encountered this species in the Sonoran Desert in northern Mexico in the 1970s. My initial interest was in foraging behavior of this ant in a desert habitat, which had not been previously described. A short natural history report (Mintzer 1979) led to initial contact with the Division of Resource Management at Organ Pipe and preliminary visits to the monument in 1980 and 1984. After the 1984 visit, resource management staff encouraged me to undertake a systematic survey. Their knowledge about colonies in the backcountry was nonexistent and indicated a clear need for a systematic survey in the park for this species to assess its population size, distribution, basic life history traits, and status. I wanted to identify several colonies in undisturbed settings that could be used for long-term study of diet, spatial pattern of foraging, and reproductive behavior. I conducted the first survey in December 1985.

Photo courtesy of Alex C. Mintzer
Natural history of A. mexicana
From their central nest that may cover up to 20 × 20 m (66 × 66 ft), colonies access an annual foraging range that may exceed 2 hectares (5 acres), using a system of lateral tunnels that extend up to 130 m (427 ft) from the nest. The workers range in size from “minims” 2 mm (~1/16 in) long to “soldiers” 12 mm (~½ in) long. The female (queen) is the largest North American ant known, with a weight of about 500 mg (0.02 oz) and a body length of 20–22 mm (~3/4–7/8 in) (fig. 4). In the course of a year, established colonies may collect about 200 kg (441 lb) (dry weight) of leaf and flower material (Mintzer 1989, 1994). All of this forage becomes substrate for the growth of the ant’s unique symbiotic fungus, cultured in large chambers deep underground under conditions of near-constant temperature and humidity. This growing fungus is the sole food source for the adults and larvae inside the nest (Weber 1972). After about a month, the fungus uses up available nutrients in the plant substrate, and the workers remove this old, exhausted fungus as waste and deposit it in large, distinctive “dump” piles on the soil surface above the central nest.

Photo courtesy of Alex C. Mintzer
Successful reproduction of A. mexicana depends upon synchronized swarming of winged reproductive (alate) ants (Mintzer 2014). Alate ants are reared every spring by mature colonies, and disperse from them with predictably precise timing in predawn darkness to mate on the morning after summer (monsoonal) storms that deliver >1 cm (0.4 in) of rainfall. Newly mated queens remove their wings and attempt to excavate a vertical burrow to escape predation and daytime heat and desiccation (Mintzer et al. 1990; Moser 1967). New colony foundation (production of the first worker offspring) requires 1–2 months (Mintzer et al. 1990; Moser 1967; Weber 1972).
Methods
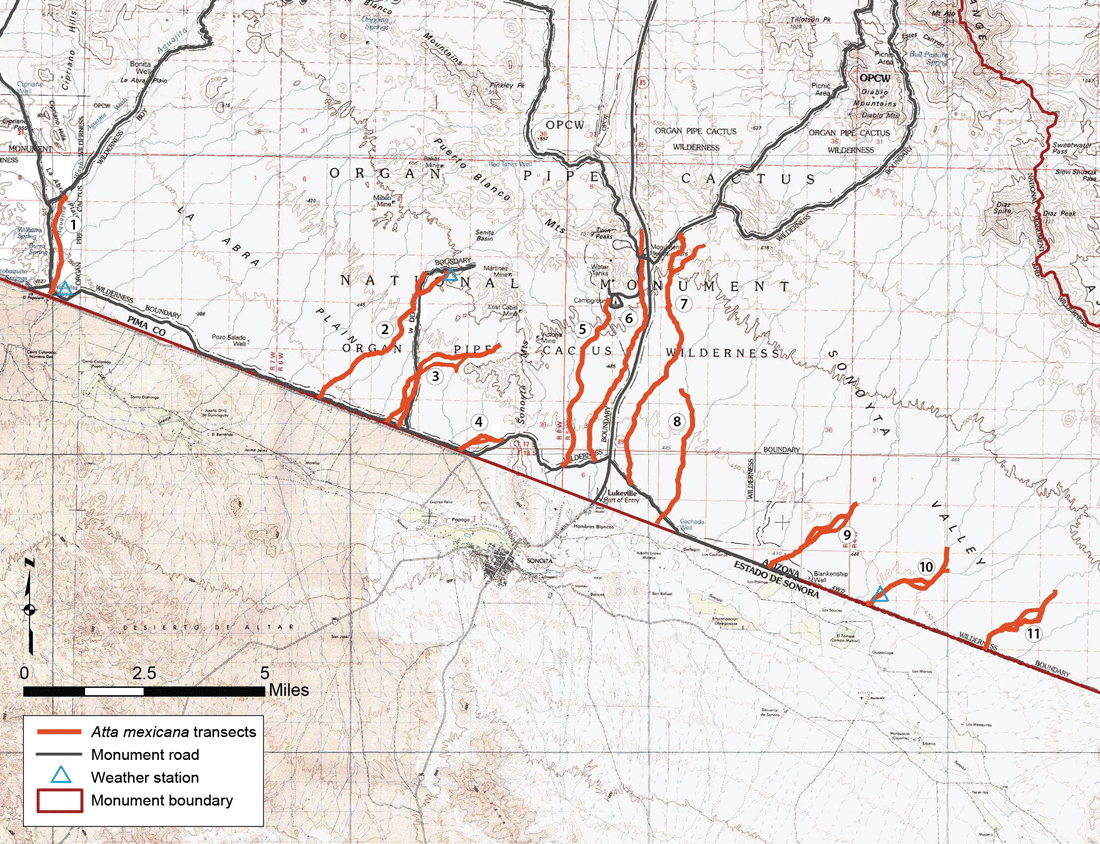
I analyzed high-resolution aerial photographs taken for Resource Management in 1976, to identify 11 large arroyo channel systems (fig. 5) in the southern one-third of the national monument with abundant vegetation to provide suitable habitat (Mintzer 1979) for these ants. I established these arroyos as transects for the repeat surveys and numbered them 1 through 11. I recruited a diverse group to assist with field searches: undergraduate and graduate student assistants, National Park Service staff, Volunteers in Parks program participants, and other motivated visitors and volunteers. Field searches employed 2–10 people spread laterally across the various banks and channels of each arroyo system; we walked the arroyo transects during daylight hours (9 a.m.–5 p.m.) when ant foraging activity was conspicuous in December or January. In addition, we noted any colonies found along the South Boundary Road extending ESE from Lukeville that provided access by vehicle to hike-in points for arroyo transects 9–11.
For each colony found, we recorded the location of the nest center in relation to the nearest arroyo channel, and any associated vegetative cover. Approximate age of individual colonies was inferred from the size and condition of superficial accumulations of excavated soil above the central nest: young colonies (see fig. 1) have fewer nest craters and smaller fungal dump deposits than middle-aged, mature nests. Growing colonies have extensive accumulations of excavated soil in neat, sharp-edged craters above their centers (figs. 1 and 6). As colonies age past maturity, the surface earthworks begin to erode away, even as the fungal dump volume and surface area continue to grow. For very old colonies, the earthworks may be completely absent (fig. 7).
The first survey was conducted in December 1985; in this pre-Global Positioning System (GPS) era, the aerial photographs proved invaluable in the field to pinpoint and map colony locations along the unique twists and braids of each channel system. In November 1986, most of these colonies were revisited and tagged with aluminum labels wired to steel rebar stakes.
Ten years later, Resource Management at Organ Pipe supported my proposal to resurvey the arroyo transects to update knowledge of ant colony distribution, population size, and life span. In January 1996, we resurveyed the park population of A. mexicana and again walked all the arroyo channel transects searched in the original 1985 survey. Although aerial photographs were again used to navigate and pinpoint colony locations, we did not physically tag the colonies located in 1996. Instead, we documented location coordinates (>150 seconds duration, with a handheld GPS instrument) as close to the colony center as possible. We used a metal detector to attempt to relocate rebar-staked tags placed at colonies, which were often concealed by silt and debris deposited by flooding events or vegetative growth in the intervening 10 years. Although it was often impossible to relocate these tags, detailed locational information from the 1985 survey and the aerial photographs allowed us to ascertain colony locations without much difficulty. In late December 2005 and early January 2006, we surveyed all the arroyo channel transects again. As in 1996, we took GPS readings as close to the colony center as possible. Finally, we surveyed the arroyo transects for a fourth time, in late December 2015 and early January 2016, using GPS field instruments. We organized the GPS readings from each day’s survey into data files and added them to the park GIS databases.

Photos courtesy of Alex C. Mintzer
Results
Table 1 provides an overview of survey results by arroyo channel transect. All 11 arroyo transects provided satisfactory habitat and hosted colonies at some point over the 30-year study period. During visits between survey periods, I also verified visitor and staff reports of backcountry colonies encountered in other parts of the park: an extinct nest along the Cuerda de Leña Wash, 2 km (1.2 mi) south of the north boundary (1987); a local cluster (deme) of four colonies in the Kuakatch wash system in the northeastern corner of the park (1989), close to the Tohono O’odham Reservation boundary; a colony near Bonita Well on the Puerto Blanco Drive and another near the first part of the Ajo Mountain Drive (early 1990s); a colony on the western park boundary (1999); a 22 km (14 mi) ground search in January 1994 along the Cherioni Wash from Highway 85 to Bates Well through the center of the park revealed only one new colony (1 km [0.6 mi] west of the highway) and no evidence of extinct nests.
| Table 1. Number of Atta mexicana colonies found along 11 arroyo transects during four surveys in the southern part of Organ Pipe Cactus National Monument, Arizona | ||||
| Arroyo/Transect | Survey Periods | |||
| 1985–1986 | 1995–1996 | 2005–2006 | 2015–2016 | |
| 1 | 0 | 0 | 0 | 3 |
| 2 | 0 | 1 | 0 | 0 |
| 3 | 1 | 1 | 0 | 1 |
| 4 | 0 | 0 | 0 | 2 |
| 5 | 0 | 1 | 2 | 0 |
| 6 | 6 | 8 | 4 | 2 |
| 7 | 7 | 8 | 6 | 8 |
| 8 | 2 | 3 | 2 | 3 |
| 9 | 0 | 4 | 3 | 21 |
| 10 | 12 | 11 | 6 | 28 |
| 11 | 8 | 8 | 2 | 15 |
| Total | 36 | 45 | 25 | 83 |
Notes: Transects are listed west to east. “Off transect” colonies along or near the international boundary not associated with these 11 transects are excluded from the totals.
1985 survey
We identified 35 living colonies in December 1985, and added two more large colonies in March 1987 along a 1 km (0.6 mi) extension of arroyo transect 6 north of the visitor center (Mintzer 1987 and 1988); this transect extension was included in all subsequent surveys.
1995–1996 survey
We located 46 living colonies along the arroyo transects in the park; one additional colony was found in the US Customs and Border Patrol inholding at Lukeville. We found colonies in seven arroyo systems and in floodplains in Senita and Lost Cabin basins. All six transects with living colonies in the December 1985 survey still had colonies in January 1996; we also found colonies along three transects that lacked colonies in 1985.
2005–2006 survey
This resurvey revealed little successful new colony recruitment and significant colony mortality over the previous 10 years. We found 25 living colonies along the transects, and one colony in Lukeville. Only four or five colonies first located in 1985 were still alive in 2005–2006, indicating that typical field colony life span is less than 20 years. We noted a substantial decline of populations west of Highway 85. We did not find any new colonies in the deme near monument headquarters, and only one colony in this area was still alive in January 2006. Unlike in previous surveys, we observed few younger colonies, and surviving colonies appeared older.
2015–2016 survey
We noted a slight population size increase in arroyo transects 1–6 to the west of Highway 85, and a major population increase in transects 7–11 east of the highway (figs. 8–10). For the first time, we found colonies in transects 1 and 4. At least nine colonies noted in 2006 were still extant in 2016, but the great majority of colonies were younger than 10 years, and most were 1–2 or 7–10 years old (table 2, page 38). In the three easternmost transects, the local density of colonies was higher than previously seen; some nests were only 30–40 m (33–44 yd) apart (fig. 10), rather than the typical 75–200 m (82–219 yd) as noted in previous surveys.
| Table 2. Number of Atta mexicana colonies with estimated colony age found along 11 arroyo transects during the 2015–2016 survey at Organ Pipe Cactus National Monument, Arizona | ||||||
| Arroyo/Transect | Estimated Colony Age (Years) | Total | ||||
| 1–2 | 2–5 | 5–7 | 7–10 | >10 | ||
| 1 | 0 | 3 | 0 | 0 | 0 | 3 |
| 2 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | 0 | 0 | 1 | 0 | 0 | 1 |
| 4 | 0 | 0 | 0 | 2 | 0 | 2 |
| 5 | 0 | 0 | 0 | 0 | 0 | 0 |
| 6 | 1 | 0 | 0 | 0 | 1 | 2 |
| 7 | 0 | 1 | 1 | 2 | 4 | 8 |
| 8 | 2 | 0 | 0 | 0 | 1 | 3 |
| 9 | 3 | 5 | 4 | 6 | 3 | 21 |
| 10 | 7 | 4 | 5 | 8 | 3 | 28 |
| 11 | 5 | 3 | 2 | 3 | 0 | 15 |
| Total | 18 | 16 | 13 | 21 | 12 | 83 |
Notes: Transects are listed west to east. “Off transect” colonies along or near the international boundary not associated with these 11 transects are excluded from the totals.
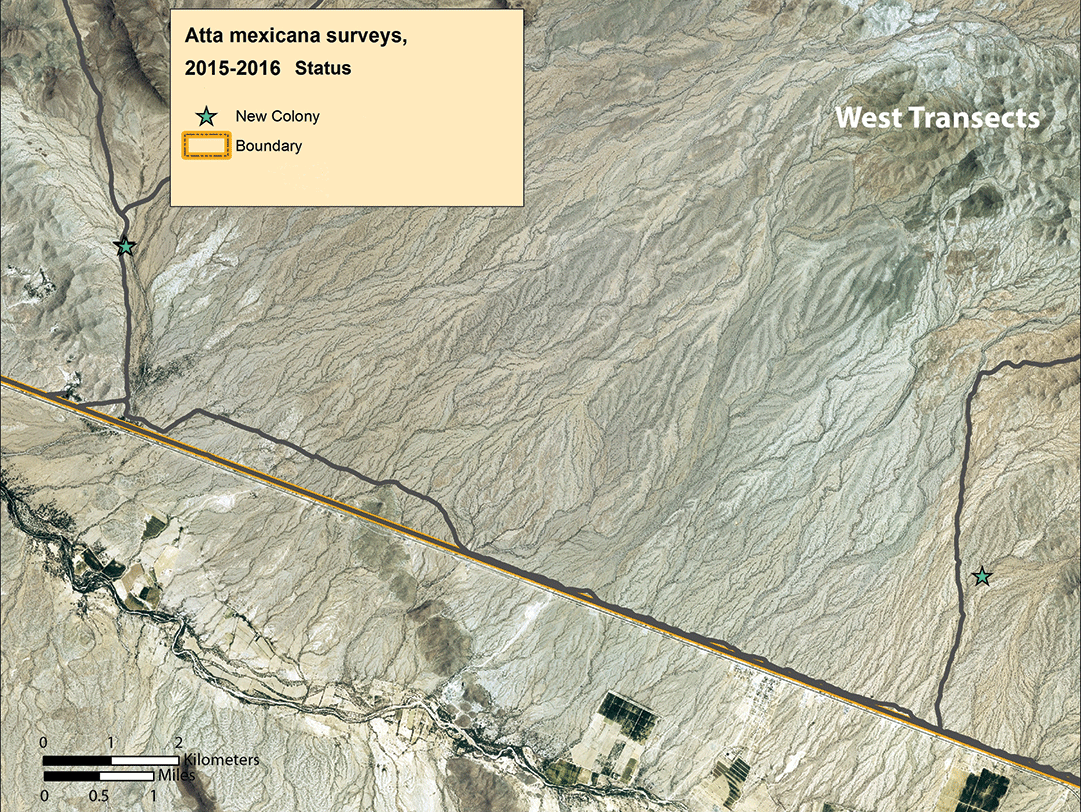
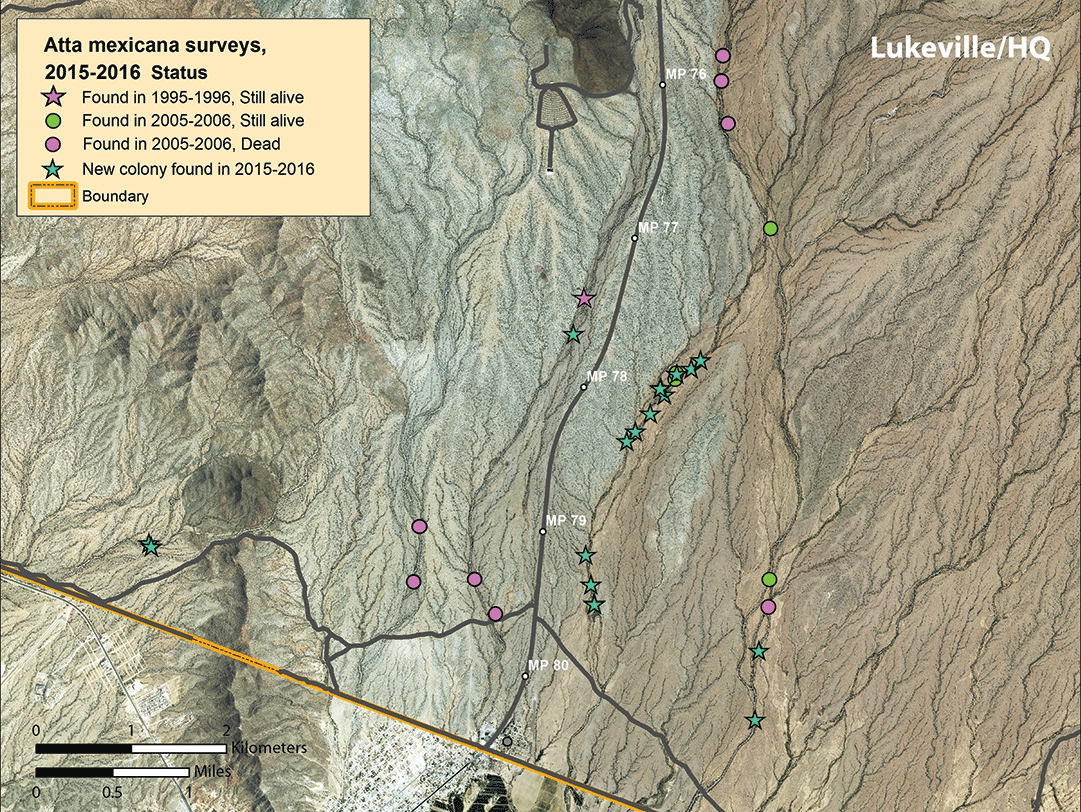
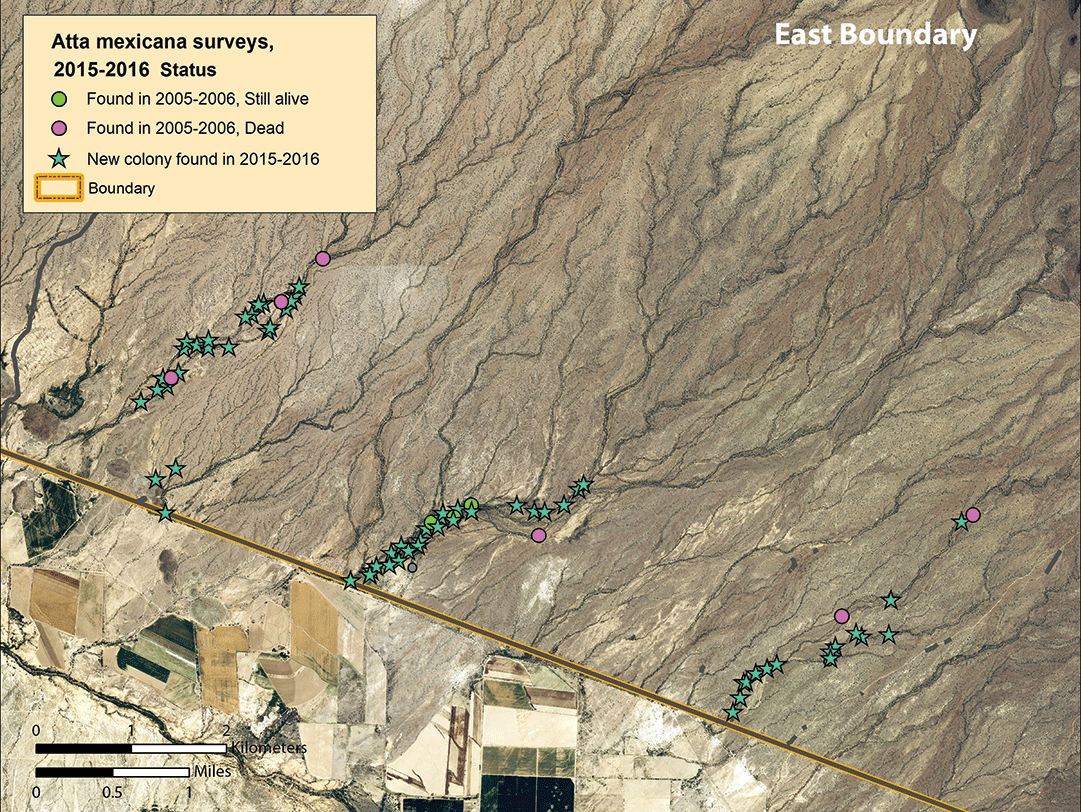
Discussion and conclusions
The A. mexicana population at Organ Pipe Cactus National Monument does not exist in a steady state: 30 years of data suggest major population size fluctuations over time (table 1). The overall spatial pattern that emerged from the four arroyo transect surveys reveals several widely scattered local clusters (subpopulations or demes) of colonies, with isolated outlier colonies as remnants of old demes undergoing local extinction, and the formation of new demes under favorable conditions (Mintzer and Mintzer 1988; Mintzer 1997, 2006).
Mean colony lifespan estimates of 10–20 years proposed in transect resurvey reports (Mintzer 1997, 2006) are consistent with estimates for other Atta species in the field and in captivity (see Weber 1972). Several A. mexicana colonies have survived at the national monument for more than 20 years, but none has survived over the complete 30-year survey period from 1985 to 2016. The deme along the northern part of arroyo system (transect) 6 near park headquarters has been followed most closely since the first survey; all five colonies present in 1987 were still alive 10 years later, but only one survived until December 2005 and it died out by October 2007. Overall, only 9 of the 26 colonies found in 2005 were still alive in 2015. At Organ Pipe, colony migration (where a colony disappearance is associated with appearance of another colony 50–200 m away) is rare; I found colonies remained in the same place during visits between the decadal surveys.
A. mexicana clearly fits within the large and growing assemblage of Sonoran plant and animal species that maintain populations with successful reproduction only in certain favorable years, and little to no success in many or most other years. Although this ant’s reproductive success varies greatly from year to year, the species has responded very positively to conditions they encountered at Organ Pipe in recent years. Population recruitment since 2005 along arroyo systems 7, 9, 10, and 11 to the east of Highway 85 is striking (figs. 9 and 10). The majority of these colonies appear to be less than seven years old, and they occur in denser local aggregations than noted in the same areas in previous surveys. The population sizes along 8 of the 11 arroyo survey transects have increased over the past 10 years, and the short-term survival outlook for the Organ Pipe population has never been more favorable.
Management implications
To a great extent, Organ Pipe Cactus National Monument has been affected by external environmental factors over the past 30 years (NPS 2013, 2016). Agricultural development, road expansion, and urbanization in the area around Sonoyta, Mexico, immediately south of the park increased greatly between 1985 and 2005 and continue to this day, threatening monument ecosystems through habitat isolation, nighttime illumination, groundwater depletion, pesticide drift, woodcutting, and invasive species facilitation (NPS 2013). Major increases in cross-border violations during this period led to an equal interdiction response by law enforcement. This included major additions to border security physical infrastructure after 2006, such as a vehicle barrier along most of the border and a 5.2-mile- (8.4 km) long pedestrian fence (NPS 2013, 2016). Additionally, long-term climate change models forecast more frequent and intense droughts in the Sonoran Desert (NPS 2016), potentially having an impact on A. mexicana and its host plant community. These anthropogenic disturbances act in concert with other biotic and abiotic factors. All of these elements have concerned resource managers at the monument for decades and led to the selection of several long-term research sites adjacent to the international park boundary in order to measure ecological changes (Bennett and Kunzman 1987; Brown 1991; NPS 1995). How these factors affect population survival and recruitment of this leafcutter ant species at Organ Pipe and thereby drive long-term changes in population size is considered in the following paragraphs in order from most to least significant.
1. Year-to-year cycles in summer precipitation. Like other Atta species (Moser 1967; Weber 1972), A. mexicana colonies at Organ Pipe do not release all their alate ants on a single night of swarming; a large number are held back for one or more subsequent nights. After heavy (5 cm [2.0 in]) rains, swarming may occur over at least two nights consecutively; after lighter rains, colonies may swarm just once and then await additional rain events over the next few weeks. While distribution of reproductive activity over multiple nights of swarming may be adaptive in habitats receiving more regular and frequent summer rains (i.e., in southern Sonora and the rest of Mexico), it carries unique risks in the Sonoran Desert.
Although monsoonal storms occur every summer at Organ Pipe, their frequency and quality vary greatly from year to year and from place to place. Since the late 1980s, the national monument’s resource management staff has accumulated daily precipitation data records from three field stations most relevant to the ant population: Aguajita to the west, Senita Basin in the center, and Salsola to the east (fig. 5). Table 3 shows the number of significant (≥1 cm and ≥1 in) summer rainfall events (June through September) recorded at these weather stations from 1995 through 2014. During at least 2 of these 20 years at Aguajita (1998 and 2002) and Senita Basin (2002 and 2011) and 1 year (2007) at Salsola, inadequate summer monsoon activity (shown in red and so noted in table 3) almost certainly prevented any swarming. For the 11 years with complete and comparable data for all three stations, no geographic west-east trend is seen for 1-inch threshold events, but the number of 1 cm (0.4 in) threshold events increased 64% from the western station (Aguajita) to the eastern station (Salsola). Not every 1 cm rain is followed by swarming; early morning (midnight to 4 a.m.) rainfall or breezes will inhibit predawn swarming, and daytime heat after modest morning rainfall may dry the soil surface and prevent swarming the following night. Thus, tabulation of 1 cm events probably overestimates the number of reproductive episodes. The 1-inch (2.5 cm) threshold, however, indicates events of higher quality that are more likely to trigger swarming over the following night or two.
| Table 3. Number of summer rainfall events for 20 years at three Organ Pipe weather stations | |||||||||
| Calendar Year |
Rainfall Events by Location
|
||||||||
|
Aguajita Wash
|
Senita Basin
|
Salsola
|
|||||||
| ≥ 1 cm | ≥ 1 in | Max (in) | ≥ 1 cm | ≥ 1 in | Max (in) | ≥ 1 cm | ≥ 1 in | Max (in) | |
| 1995 | ND | ND | ND | 3 | 1 | 1.11 | 6a | 2a | 2.39a |
| 1996 | 3 | 1 | 1.09 | 4 | 1 | 1.21 | 6a | 2a | 2.45a |
| 1997 | 1b | 0b | 0.70b | 3 | 0 | 0.76 | 5a | 1a | 1.01a |
| 1998 | 0c | 0c | 0.26c | 3 | 0 | 0.85 | 4 | 2 | 1.54 |
| 1999 | ND | ND | ND | 3 | 0 | 0.78 | 6a | 0a | 0.91a |
| 2000 | ND | ND | ND | 3 | 0 | 0.79 | 4 | 0 | 0.85 |
| 2001 | ND | ND | ND | 4 | 0 | 0.75 | 2 | 1 | 1.23 |
| 2002 | 0c | 0c | 0.15c | 0c | 0c | 0.25c | 2b | 0b | 0.46b |
| 2003 | 6a | 2a | 1.87a | 4 | 3 | 2.11 | 5a | 2a | 1.48a |
| 2004 | ND | ND | ND | 2b | 0b | 0.92b | 1b | 0b | 0.93b |
| 2005 | 2 | 1 | 1.32 | 3 | 1 | 1.15 | 1b | 0b | 0.59b |
| 2006 | 3 | 0 | 0.74 | 2b | 0b | 0.60b | 5a | 1a | 1.70a |
| 2007 | 1 | 1 | 2.33 | 2b | 0b | 0.79b | 0c | 0c | 0.35c |
| 2008 | 1 | 1 | 1.28 | ND | ND | ND | 4 | 2 | 2.06 |
| 2009 | 1b | 0b | 0.49b | 0? | 0? | ND | 1b | 0b | 0.78b |
| 2010 | ND | ND | ND | 1 | 1 | 1.14 | 1b | 0b | 0.65b |
| 2011 | ND | ND | ND | 0c | 0c | 0.39c | 3 | 1 | 2.82 |
| 2012 | 7a | 4a | 1.50a | 6a | 1a | 1.08a | 8a | 0a | 0.92a |
| 2013 | 3 | 1 | 1.39 | 3 | 1 | 1.23 | 5a | 1a | 3.92a |
| 2014 | 2b | 0b | 0.80b | 3 | 0 | 0.76 | 5a | 1a | 1.75a |
| Cumulative | 28 | 10 | 33 | 7 | 46 | 10 | |||
Note: Rainfall events are from daily records over four months from June through September. Maximum daily rainfall is shown during these periods. Cumulative counts (last row) only include 11 years when comparable complete records are available for all three stations.
ND indicates station records with missing data.
aGreen denotes wetter years.
bBrown signifies drier years.
cRed indicates the driest years.
Successful reproduction and population recruitment of new colonies appear to be associated with summers with more rain events, heavier maximum rain events, or both. Such summers provide leafcutter ant colonies with ample opportunities to deploy all or nearly all of their alates and maximize their reproductive success. Newly founded nests may survive the late summer and fall best in wet years. The Salsola weather station is located within arroyo transect 10, which had more Atta colonies than any other transect during three of the four surveys (and tied with transect 7 for most colonies in the 2005–2006 survey; see table 1). Salsola also provides an uninterrupted daily rainfall record over 20 years (1995–2014; table 3). Over the 10-year period 2005–2014, six years (2006, 2008, 2011, 2012, 2013, and 2014) delivered above-average frequency of 1 cm threshold events, heavier maximum rainfall events, or both. The age data (table 2) for transect 10 show a diverse range of colony ages, indicating successful reproduction during multiple “good” years spread throughout the decade, although it is not possible to ascribe origins of any specific colony to a particular rain event.
2. Anthropogenic climate change probably is an important factor, especially for the long-term future. These ants are living at their limits of tolerance for arid conditions. The American Southwest in general has experienced more frequent drought conditions in recent decades. Consensus climate change models forecast intensification of this trend this century (NPS 2016). The timing and quantity of summer and winter rainfall determine the health of host plants of the ant and the availability of foliage, flowers, and fruits for harvest. In the Sonoran Desert, A. mexicana colonies along arroyo channel margins have access to a mix of important host plants: larger trees such as palo verde (Cercidium spp. and Parkinsonia spp.), ironwood (Olneya spp.), mesquite (Prosopis spp.), and catclaw (Acacia spp.) along the channels, and creosotebush (Larrea spp.) in the open desert away from these channels. The ants depend upon these plants throughout the year. In addition, increased shade and soil moisture along arroyo channels is probably vital for new colony foundation and population recruitment. Along the western transects, diversity and coverage of larger trees (except Prosopis) is reduced, and abundant saltbush/bursage (Franseria spp./Ambrosia spp.) populations reduce the density of Larrea in the open desert. A. mexicana populations are smaller and more isolated in the western transects at Organ Pipe, which experience lower mean annual rainfall. For the 11 most recent years with complete and comparable data, mean annual rainfall was 6.09 ± 2.51 inches (15.47 ± 6.38 cm) at Aguajita (at the western edge of the survey area), 7.22 ± 2.82 inches (18.34 ± 7.16 cm) at Senita Basin, and 9.02 ± 2.70 inches (22.91 ± 6.85 cm) at Salsola along densely populated arroyo transect 10 to the east. Extended drought conditions that eliminate threshold (1 cm [0.4 in]) monsoonal rainfall events over a decade or more would prevent swarming activity, prevent successful colony establishment, and lead to decline or extinction of local populations. Sadly, A. mexicana should be a good indicator species for effects of sustained severe drought at Organ Pipe.
3. Inferred potential decreases in local populations or effectiveness of natural alate ant predators, particularly bats, nighthawks, and Colorado River toads. The large size and innocuous nature of alate A. mexicana males and females make them attractive food items for local nocturnal and crepuscular (dawn-active) insectivores, and it was easy to observe predation by bats and toads associated with predawn swarming activity in 1986, 1987, 1989, and 1997 at Organ Pipe. Even though Atta alates are a rare food resource, available in abundance only a few nights a year and in very limited areas, bats were seen circling and gleaning alates on the ground and those taking flight from swarming colonies. Nighthawks are also common at Organ Pipe and are a known predator of alates of other Atta species (Moser 1967). Other opportunistic mammalian predators that may seek alates and young colonies at Organ Pipe include the American badger and spotted skunks; armadillos, observed to feed on A. texana in Louisiana (Moser 1967), are absent in Arizona.
Comparison of Salsola rainfall data (1995–2004 versus 2005–2014 in table 3) does not show a convincing decadal trend to explain overall increased reproductive success since 2005, so we must consider alternative explanations. The more recent surge in number of colonies in transects 9–11 near Salsola suggests that predator pressure on this local ant subpopulation may have decreased. This could be due to reduced predator population sizes, changes in predator foraging behavior, or temporary satiation of local predators associated with dense Atta swarms.
4. Increased human population and agricultural land use in Sonoyta, Mexico, adjacent to the national monument could isolate Organ Pipe populations of A. mexicana from source populations to the south, but this did not prevent vigorous colony reproduction and population recruitment over the last four years. These numerous younger colonies were almost certainly founded by females originating locally from the few surviving mature colonies in Organ Pipe or from colonies within a few kilometers of the international boundary. Clearance of land for agriculture that isolates park populations of A. mexicana from source populations in Mexico would have long-term effects on genetic diversity. The alates are not strong fliers and only swarm on calm nights; dispersal distances of winged reproductive Atta females are probably less than 5 km (3.1 mi) (Moser 1967). Very slight airflow from the southeast associated with monsoonal conditions may have had a role in moving some alate females from borderland Sonora into Organ Pipe.
There is no evidence so far suggesting that pesticide drift and groundwater depletion associated with intensive agriculture have affected host plants and ant populations near the international boundary. Cutting of Olneya and Prosopis trees in riparian habitats near the boundary could also have an effect. Because alate ants are attracted to lights, nighttime illumination will affect dispersal of some winged reproductive ants during predawn hours. However, these ants continue to thrive in well-lighted, urbanized areas in central and coastal Mexico, so this also may be a minor concern.
5. Increased border security infrastructure has the potential to amplify effects outlined in the factor above. Construction of a vehicle barrier along the international boundary began in 2003, but the limited vegetation clearance and 2-meter- (6.6 ft) deep posthole excavation associated with it have not had a major impact on colonies along the southern monument boundary. However, construction of the 18-foot pedestrian barrier (fence), widening of roads, and installation of high-intensity nighttime illumination along limited areas of the border began in 2007, and this infrastructure could have a negative effect on A. mexicana populations at Organ Pipe. Also, the use of all-terrain vehicles to interdict cross-border violators has produced visible local degradation of near-border plant communities (NPS 2013), although the effects on colony survival may be minimal for reasons outlined in the fourth factor above.
6. Cross-border activity is another factor. This activity increased significantly between 1985 and 2005 at Organ Pipe (NPS 2013), and was often concentrated along the large arroyo channel systems that are preferred habitat of A. mexicana in the Sonoran Desert. Since 2008, cross-border violations have decreased significantly, though certain routes, like arroyos, tend to be used with some frequency and routes change often in response to interdiction (NPS, T. Tibbitts, resource management specialist, personal conversation, April 2012). Transient pedestrian traffic probably has negligible effects on ant foraging behavior, and accumulation of trash probably has no effect on established colonies. Cross-border violators often break off lower branches to open up sheltering spaces under some larger trees along arroyos, but as long as these large host trees (Parkinsonia, Cercidium, Olneya, Prosopis, Acacia) are not seriously damaged by transient activity, this impact will be minor; in any case, the deep nests are highly resistant to local disturbance.
Conclusion
Atta mexicana is a charismatic invertebrate with complex social behavior and fascinating natural history, living at the limits of its tolerance for aridity and temperature extremes. Like other such species surviving at the margins of their range, it should be a sensitive indicator for long-term environmental impacts. Although Resource Management has not yet included this ant in its long-term monitoring program, its support for decadal surveys is likely to continue. Finally, an effort should be made to integrate molecular genetics technology with future study of this park population to assess inbreeding, genetic diversity levels (always a concern in small, isolated populations), gene flow (effective dispersal), colony genetic structure (number of queens per colony), and to estimate effective population size.
Acknowledgments
Many individuals assisted with field surveys: my spouse Carole; National Park Service staff Terry Peters, Bill Mikus, Dick and Margaret Anderson, Charles Conner, Tim Tibbitts, Ami Pate, Nancy Reid, Lara Dickson, and Willie Rohde; volunteers Ruth and “Freddie” Fredericks, Steve Prchal, Chip Hedgcock, Vaughn Tippit, Steve Birt, and Elna Otter; Terry Marsh and his students; student assistants Kevin Haack, Ahmar Hashmi, Rebecca Clark, Tate Holbrook, Steven Koteras, and Andrew Alton. Ami Pate prepared the transect maps. This research was supported by grants from Southwest Parks and Monuments Association (Tucson, Arizona) and Western National Parks Association (Tucson, Arizona).
References
Bennett, P. S., and M. R. Kunzman. 1987. Organ Pipe Cactus National Monument Biosphere Reserve Sensitive Ecosystems Program. Special Report Number 7. Cooperative National Park Resources Studies Unit, University of Arizona, Tucson, Arizona, USA. 82 pages.
Brown, B. 1991. Land use trends surrounding Organ Pipe Cactus National Monument. Technical Report No. 39. Cooperative National Park Resources Studies Unit, University of Arizona, Tucson, Arizona, USA. 65 pages.
Byars, L. F. 1949. The Mexican leaf-cutting ant in the United States. Journal of Economic Entomology 42:545.
Creighton, W. S. 1950. The ants of North America. Bulletin of the Museum of Comparative Zoology 103. 585 pages.
Hölldobler, B., and E. O. Wilson. 1990. The ants. Belknap Press, Harvard University, Cambridge, Massachusetts, USA. 732 pages.
Mintzer, A. C. 1979. Foraging activity of the Mexican leafcutting ant Atta mexicana (F. Smith) in a Sonoran Desert habitat (Hymenoptera, Formicidae). Insectes Sociaux 26:364–372.
———. 1987. The status of the leafcutting ant Atta mexicana at Organ Pipe Cactus National Monument. Final Project Report. Southwest Parks and Monuments Association, Tucson, Arizona, USA. 15 pages.
———. 1989. Diet and foraging behavior of the leafcutting ant Atta mexicana at Organ Pipe Cactus National Monument. Final Project Report. Southwestern Parks and Monuments Association, Tucson, Arizona, USA. 19 pages.
———. 1994. Diet of the leafcutting ant Atta mexicana in a Sonoran Desert habitat. Proceedings of the Arizona-Nevada Academy of Sciences 28:33–40.
———. 1997. Population status of the leafcutting ant Atta mexicana at Organ Pipe Cactus National Monument: A 10-year resurvey of wash habitats. Final Project Report. Southwest Parks and Monuments Association, Tucson, Arizona, USA. 6 pages.
———. 2006. Changes over 20 years in populations of the Mexican leafcutting ant Atta mexicana at Organ Pipe Cactus National Monument, Arizona. Final Project Report. Western National Parks Association, Tucson, Arizona, USA. 13 pages.
———. 2014. Atta mexicana swarming activity at Organ Pipe Cactus National Monument, AZ. YouTube video (5 minutes). https://www.youtube.com/watch?v=1tYroFc3QyA.
Mintzer, A. C., and C. L. Mintzer. 1988. Population status of the Mexican Leafcutting ant, Atta mexicana (Formicidae) in the Sonoran Desert of Arizona. The Southwestern Naturalist 33:250–251.
Mintzer, A. C., C. Deloya, and L. Quiroz. 1990. Foundation of colonies of the ant Atta mexicana (Hymenoptera: Formicidae) in the laboratory (English). Folia Entomología Mexicana 82:133–138.
Moser, J. C. 1967. Mating activities of Atta texana. Insectes Sociaux 14:295–312.
NPS (National Park Service). 1995. Organ Pipe Cactus National Monument Ecological Monitoring Program protocol manual. Special Report No. 11. National Biological Service Cooperative Park Studies Unit, University of Arizona, Tucson, USA.
———. 2013. State of the Park report for Organ Pipe Cactus National Monument. State of the Park Series No. 3. National Park Service, Washington, DC, USA. 47 pages.
———. 2016. Foundation document. Organ Pipe Cactus National Monument, Washington, DC, USA. 60 pages.
Smith, M. R. 1963. Notes on the leaf-cutting ants, Atta spp., of the United States and Mexico. Proceedings of the Entomological Society of Washington 65:299–302.
Weber, N. A. 1972. Gardening ants, the attines. Memoirs of the American Philosophical Society, vol. 92. Philadelphia, Pennsylvania, USA. 146 pages.
Wheeler, G. C., and J. Wheeler. 1983. The superstructures of ant nests. Transactions of the American Entomological Society 109:159–177.
About the author
Alex C. Mintzer (amintzer@cypresscollege.edu or alexmintzer@socal.rr.com) is a professor of biology at Cypress College, Cypress, California.
