Part of a series of articles titled Alaska Park Science - Volume 19, Issue 1 - Below the Surface: Fish and Our Changing Underwater World.
Article
Eulachon: A Little Fish with Many Names and a Big Ecological Impact
Andrew Waldo, Alaska Department of Fish & Game
Jami Belt, National Park Service

NOAA FISHERIES
Each spring in Southeast Alaska, people and wildlife eagerly await the same phenomenon: silvery schools of a small nutrient-rich fish. Called hooligan or candlefish by Alaskans, but referred to as eulachon by biologists, Thaleichthys pacificus is an anadromous smelt. Southeast Alaska spawning populations spend most of their life feeding and growing in cold North Pacific waters before migrating to rivers during spring to reproduce. One of these rivers is the Taiya, which flows from the Coast Mountains, through Klondike Gold Rush National Historical Park (NHP), finally spilling into an estuary near Dyea, a former gold rush town. Although Taiya River eulachon fueled generations of people and wildlife, we know little about them or their current population status. Such information would be valuable since southern populations are in decline and the Taiya River has hydroelectric potential, which could impact the species (Johnson and Twenhofel 1958, Municipality of Skagway 2014).
Eulachon once ranged from northern California to the Alaska Peninsula and southeast Bering Sea (Brown et al. 2005, Gustafson et al. 2012); today southern runs are severely depleted or extinct. During 2010, the population segment that spawns in California, Oregon, and Washington was listed as threatened under the Endangered Species Act (NOAA 2010) while spawning populations south of the Nass River in British Columbia are of “special concern” (BC Conservation Data Centre 2017). Changes in ocean conditions, declines in quantity and quality of spawning habitat, and overfishing are all linked to eulachon declines (Gustafson et al. 2012). Because the species is not commercially harvested, basic data is lacking for most populations even though they fueled West Coast Native cultures and trade for generations.
Oil, medicine, food, and trade items provided by this diminutive fish influenced generations of indigenous peoples leading anthropologists to consider eulachon a keystone species of West Coast Native culture (Senkowsky 2007). Since eulachon runs occur when human food supplies historically ran low, some tribes called them “salvation fish” (MacKinnon 2015). High in oils, up to 20% of the eulachon mass is fat (Wilson et al. 2006) and just 150 ml (5 oz) provides half an adult human’s daily energy needs (MacKinnon 2015). The buttery oil is produced by fermenting large quantities of whole fish and then extracting the resulting nutrient-dense oil. The oil is added to other foods or consumed alone for medicinal purposes. The fish itself is prized as a delicacy and when dried it can be ignited for a light source, hence the name candlefish. The famous Chilkoot Trail’s history reflects the species importance. During the 1890s gold rush, about 30,000 individuals (Karl Gurcke, pers. comm. 2016) traversed the Chilkoot across the rugged Coast Mountains on their way to the Klondike Gold Fields. However, before the gold rush, generations of Alaska Natives used the famous Chilkoot Trail and similar trails to trade eulachon oil with their interior neighbors, such trails were nicknamed “grease trails” (Flannery et al. 2009). Today subsistence fishers harvest eulachon throughout Southeast Alaska (Brown et al. 2005). Little is known regarding the extent of eulachon subsistence use in the Taiya River or Skagway, but nearby in Haines and Klukwan, 95% of households reported using eulachon and subsistence fishers reported a harvest of 162,060 pounds during 2012 (ADFG 2020).
Not only do eulachon sustain human cultures, but this small silvery-blue fish also supports diverse Alaskan wildlife predators. Spawning runs in Southeast Alaska typically arrive at estuaries and rivers in early spring, a time of high energy demand for sea lions, river otters, harbor seals, and migrating shorebirds, all of which feast on eulachon (Sigler et al. 2004). Sea lions haul out near eulachon migration paths (Womble et al. 2005), and the little energy-dense fish helps sustain the massive pregnant females and migrating males. Whales, dolphins, sharks, bears, wolves, ravens, eagles, seabirds, salmon and many other species exploit eulachon when available (Willson et al. 2006). A eulachon study by Marston and others (2002) indicated that “predator abundance rose rapidly at the beginning of the runs and was significantly correlated with an index of eulachon abundance within years.” Since eulachon return to Southeast Alaska when other prey is scarce, they are considered an ecological cornerstone for coastal systems (Marston et al. 2002).
Like salmon, most eulachon are semelparous, meaning reproduction is fatal-fish die soon after a single spawning event (Clarke et al. 2007). Eulachon generally spawn during spring in lower river reaches once they reach age 2-5 and are about as long as a pencil (6.3-9.8 inches or 160-250 mm; fork length—tip of nose to fork of tail). Females produce 7,000-60,000 eggs which are broadcast over gravel or sand (Willson et al. 2016). The sticky fertilized eggs attach to substrate and hatch in 3-8 weeks depending on water incubation temperature; embryos develop and hatch faster in warmer waters and slower in cooler waters (Willson et al. 2006). Eulachon spawn in different rivers and regions across a wide range of river temperatures (32o-50oF or 0o-10oC) including under ice (Willson et al. 2006, Gustafson et al. 2012).
Estimates suggest fewer than 60 Alaskan rivers host anadromous eulachon runs (Willson et. al 2006). While studies exist for some southeast Alaska rivers, research is lacking for the park’s Taiya River. The Chilkoot Indian Association and the Takshanuk Watershed Council studied eulachon population trends in the nearby Chilkoot River during 2010-2016. Based on their success, park biologists, the Skagway Traditional Council, and the Taiya Inlet Watershed Council initiated the first study of eulachon in the Taiya River in an effort to gather basic population information.

PHOTOS COURTESY OF SARAH APSENS, ALASKA SEAGRANT STATE FELLOW
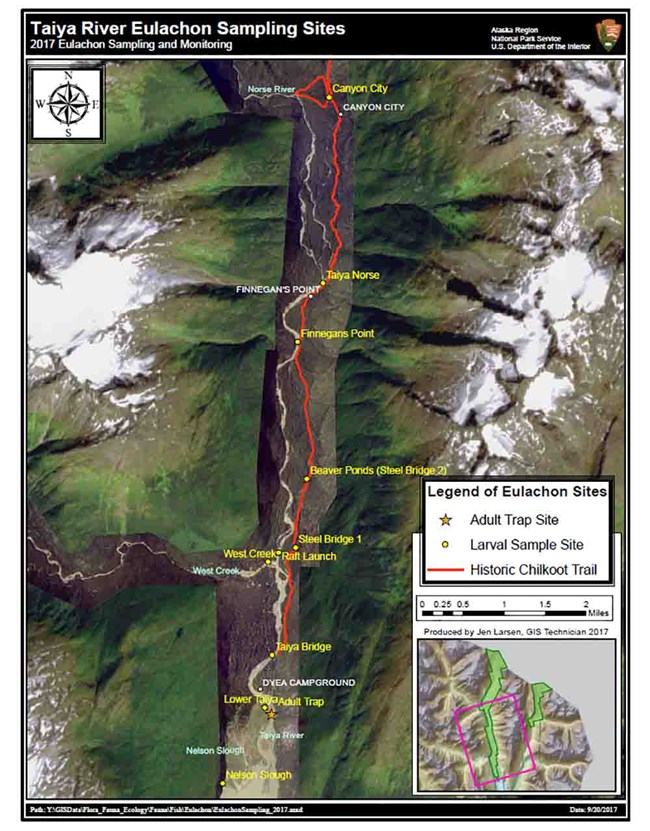
During 2017 and 2018, biologists sampled Taiya River eulachon run timing, relative abundance, sex ratios, adult weight and length, female fecundity, and larval distribution. Biologists installed a fyke net in mid-April, to sample upriver migrating adults. Trapping occurred during daylight, five days a week, for one hour each incoming, outgoing, and slack tide. Captured fish were counted, sexed, weighed, measured then returned to the river. A subset of females was sacrificed to determine fecundity. Presence of sea lions, an indicator of eulachon run strength (Marston et al. 2002), was noted prior to daily sampling. Sampling for out-drifting eulachon larvae and eggs as an indicator of spawner distribution and habitat began three weeks after the first adult eulachon were captured; sampling occurred weekly at 10 different sites (Figure 1).
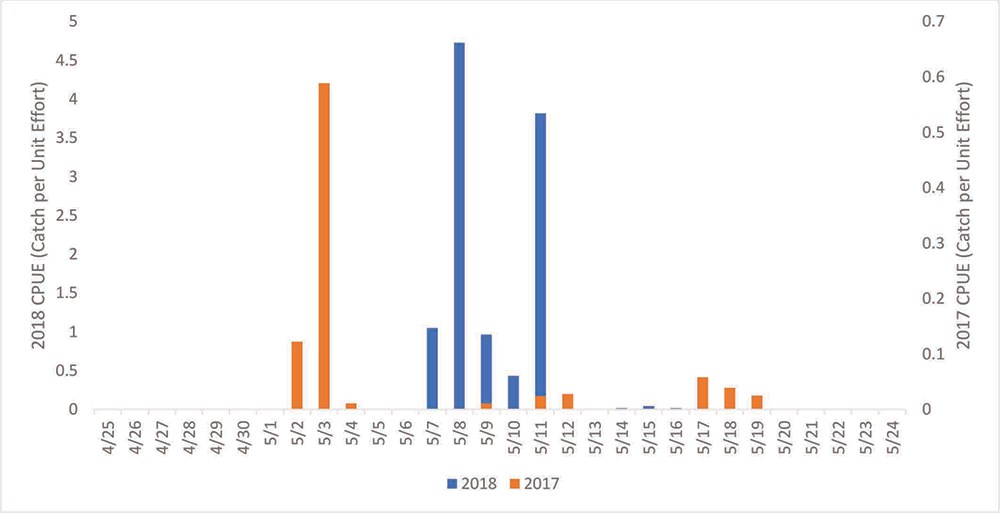
During both study years, the Taiya River spawning run began in early May and lasted approximately one week in 2017 and approximately two weeks in 2018. Catch Per Unit Effort (CPUE) indicated low abundance both years with peak catch being 0.06 fish/minute in 2017 and 4.7 fish/minute in 2018 (Figure 2). Low eulachon abundance was also reflected in the number of sea lions observed. One sea lion was observed in 2017 and none in 2018; in contrast, 75 sea lions were observed at the study site during 2016 (Jami Belt, personal communication, 2016) indicating a larger eulachon run. It is not possible to draw conclusions regarding Taiya River eulachon abundance trends from a two-year study and past studies show some eulachon runs are reliable from year to year while others are sporadic (Willson et al. 2006). Studies of total eulachon abundance in the nearby Chilkoot River showed wide fluctuations during 2010-2016, ranging from 319,586 to 12.6 million fish, with more than a million fish documented in five of the six study years (CIA 2017). Because census of eulachon abundance requires significant human and financial capital—both limited for Klondike Gold Rush NHP—the need for and means to obtain such information should be carefully considered. In addition to standard census techniques, the utility and cost of advancing genetic tools such as eDNA, SNPS, and microsatellites should be evaluated if abundance estimates are again considered (see Simmons et al. this issue, Candy et al. 2015, Carraro et al. 2018). Pochardt and others (2020) tested an eDNA approach for estimating abundance during spawning using 2017 eDNA samples from the Taiya River that yielded promising results.
Tayia River eulachon size, sex ratio, and fecundity varied between study years. These traits are important since they can provide clues regarding eulachon population health and trends. For example, changing environmental conditions such as high temperatures or reduced food supply can stress fish contributing to higher fish mortality and selecting for a younger, smaller, less fecund reproductive population (Heino and Kaitala 1999, Sutherland et al.1986). During the 2017 Taiya River study, both sexes were similar in size averaging 1.1 oz (about 32.1 g) and 6.7 inches long (about 170.3 mm); in 2018, females were smaller than males, and smaller than in 2017, averaging 0.9 oz (25.5 g) and 6.2 inches (157.9 mm). Males averaged 1.1 oz (29.9g) and 6.5 inches (166.0 mm) similar to 2017. Female fecundity in 2017 averaged 16,012.12 eggs per female (range=10,798.99-27,798.55) 2017 and in 2018 averaged 16,553.03 eggs per female (range=2,436.94-26,169.18). These results are comparable to observations of other Southeast Alaska eulachon populations (Willson et al. 2006) and provide the first life history baseline information for the Taiya River eulachon.
No larvae or eggs were observed in or above West Creek during the two-year study, suggesting spawning did not occur in those areas. Uppermost presence of eulachon documented in both years was at the bridge south of the Beaver ponds, approximately five river miles from the estuary and well above tidal influence contrary to numerous studies indicating eulachon only spawn in tidally influenced reaches. Our results provide some basic information on eulachon spawning distribution in the Taiya River which can aid future conservation of essential habitat should hydroelectric or other development occur.
Taiya River eulachon face an uncertain future. Similar to other high-latitude seas, the Gulf of Alaska is predicted to become more acidic and warmer in the future (Fabry et al. 2009). Ocean acidification may impact distribution and abundance of eulachon prey, while warming water effects include: increased fish metabolism and changes to normal growth and maturation rates; changes in spawn run timing; changes in egg incubation rates, and changes in mortality rates, to name a few. This study provides the first brief glimpse into the abundance and characteristics of Taiya River eulachon. Their persistence will likely be tied to their ability to adapt to future changes and conservation of their essential habitats.
To learn more about this project, check out this short video about the eulachon monitoring project in Klondike Gold Rush National Historical Park.
References
Alaska Department of Fish & Game (ADFG). 2020.
Community Subsistence Information System: CSIS. Available at: https://www.adfg.alaska.gov/sb/CSIS/ (accessed March 20, 2020)
BC Conservation Data Centre. 2017.
BC Species and Ecosystems Explorer. BC Ministry of the Environment Victoria, B.C. Available at: http://a100.gov.bc.ca/pub/eswp/ (accessed December 12, 2017)
Brown, C. L., D. Caylor, J. Dizard, J. A. Fall, S. Georgette, T. Krauthoefer, and M. Turek. 2005.
Alaska subsistence salmon fisheries 2003 annual report. Alaska Department of Fish and Game, Division of Subsistence, Technical Paper No. 316. Juneau, AK.
Candy, J. R., N. R. Campbell, M. H. Grinnell, T. D. Beacham, W. Larson, S. R. Narum. 2015.
Population differentiation determined from putative neutral and divergent adaptive genetic markers in eulachon (Thaleichthys pacificus, Osmeridae), an anadromous Pacific smelt. Molecular Ecology Resources 15: 1421–1434.
Carraro, L., H. Hartikainen, J. Jokela, E. Bertuzzo, and A. Rinaldo. 2018.
Estimating species distribution and abundance in river networks using environmental DNA. Proceedings of the National Academy of Sciences 115 (46): 11724-11729.
Chilkoot Indian Association (CIA). 2017.
Identifying climate vulnerabilities and prioritizing adaptation strategies for eulachon populations in the Chilkoot and Chilkat River and the application of local monitoring systems. Prepared for North Pacific Landscape Conservation Cooperative & Alaska Climate Science Center. Agreement number F13AP01047.
Clarke, A. D., A. Lewis, K. H. Telmer, and J. M. Shrimpton. 2007.
Life history and age at maturity of an anadromous smelt, the eulachon Thaleichthys pacificus (Richardson). Journal of Fish Biology 71: 1479-1493.
Fabry, V. J., J. B. McClintock, J. T. Mathis, and J. M. Grebmeier. 2009.
Ocean acidification at high latitudes: The bellwether. Oceanography 22(4): 160-171.
Flannery, B. G., J. K. Wenburg, C. J. Lewis, B. L. Norcross, and R. E. Spangler. 2009.
Genetic population structure of Alaska eulachon. U.S. Fish and Wildlife Service. Alaska Fisheries Technical Report Number 106.
Gustafson, R. G., M. J. Ford, P. B. Adams, J. S. Drake, R. L. Emmett, K. L. Fresh, M. Rowse, E. A. K. Spangler, R. E. Spangler, D. J. Teel, M. T. Wilson. 2012.
Conservation status of eulachon in the California Current. Fish and Fisheries 13: 121-138.
Heino, M. and V. Kaitala. 1999.
Evolution of resource allocation between growth and reproduction in animals with indeterminate growth. Journal of Evolutionary Biology 12:423-9.
Johnson, A. and W. S. Twenhofel. 1958.
Potential industrial sites in the Lynn Canal area, Alaska. Geological Survey circular 280. Washington D.C. 17 pp. Available at: https://pubs.usgs.gov/circ/1953/0280/report.pdf (accessed March 20, 2020)
MacKinnon, J. B. 2015.
“Salvation fish” that sustained native people now needs saving. National Geographic. July 7, 2015. Available at: https://www.nationalgeographic.com/news/2015/07/150707-salvation-fish-canada-first-nations-animals-conservation-world/ (accessed March 20, 2020)
Marston, B. H., M. F. Willson, and S. M. Gende. 2002.
Predator aggregations during eulachon Thaleichthys pacificus spawning runs. Marine Ecology Progress Series 231: 229-236.
Municipality of Skagway. 2014.
West Creek Master Plan. Skagway ordinance no. 14-09. Available at: https://www.skagway.org/sites/default/files/fileattachments/community/page/28411/july_2014_
approved_west_creek_master_plan_web.pdf (accessed March 20, 2020)
National Oceanic and Atmospheric Administration (NOAA). 2010.
Endangered and threatened wildlife and plants: threatened status for southern distinct population segment of eulachon. Federal Register 75(52).
Pochardt, M., J. M. Allen, T. Hart, S. D. K. Miller, D. W. Yu, and T. Levi. 2020.
Environmental DNA facillitates accurate, inexpensive, and multiyear population estimates of millions of anadromous fish. Molecular Ecology Resources 20: 457-467.
Senkowsky, S. 2007.
A feast to commemorate—and mourn—the eulachon. BioScience 57 (8):720.
Sigler, M. F., J. M. Womble, and J. Vollenweider. 2004.
Availability to Steller sea lions (Eumetopias jubatus) of a seasonal prey resource: a pre-spawning aggregation of eulachon (Thaleichthys pacificus). Canadian Journal of Aquatic Science 61:1475-1484.
Sutherland, W. J., A. Orafen, and H. P. Harvey. 1986.
Life history correlations and demography. Nature 320 (6057): 88.
Willson, M. F., R. H. Armstrong, M. C. Hermans, and K.Koshki. 2006.
Eulachon: A review of biology and an annotated bibliography. Alaska Fisheries Science Center, Processed Report 2006-12.
Womble, J. N., M. F. Willson, M. F. Sigler, B. P. Kelly, and G. R. VanBlaricom. 2005.
Distribution of Steller sea lions Eumetopias jubatus in relation to spring-spawning fish in southeast Alaska. Marine Ecology Progress Series 294: 271-282.
Last updated: May 29, 2020
