Part of a series of articles titled Alaska Park Science - Volume 17, Issue 1. Migration: On the Move in Alaska.
Article
History, Purpose, and Status of Caribou Movements in Northwest Alaska

NPS /Kyle Joly
Caribou (Rangifer tarandus) are thought to be a species of Eurasian descent (Osborn 1910). While the species is known for having high fidelity to their calving grounds, the establishment of new calving grounds was essential to allow the species to expand its distribution, which now spans the Arctic globally. Flexibility in this behavioral trait allowed for eastward expansion, hundreds of thousands of years ago, across the now-submerged Bering Land Bridge (Banfield 1962, Guthrie and Matthews 1971).
People followed caribou across the land bridge much more recently, perhaps some 15,000 years ago. These first Alaskans relied on caribou for food, clothing, and tools, and the species has played a prominent role in Alaska Native culture for thousands of years (Anderson 1968, Burch 1972). People who depend on caribou are keenly aware of their movements and have needed to be mobile and flexible enough to move to where the caribou are or were heading. As Noorvik elder Clarence Jackson recalls, he would “hunt way up to the head of the Noatak River, stay there ten days to two weeks until they had enough caribou to bring home” (Betcher 2016). Entire families would work together to drive caribou toward waiting hunters during migration. These community caribou drives are no longer conducted for numerous reasons, including the adoption of new technologies, such as the use of firearms and motorized transportation (Burch 2012). However, Alaska Natives continue to harvest caribou during their migrations by anticipating and then intercepting their movements at strategic locations using knowledge that has been passed down through generations.
Migration is a distinctive behavioral trait of caribou. Caribou display the longest terrestrial migrations anywhere on the planet. In northwest Alaska, caribou travel up to 2,737 miles (4,404 km) per year (Joly and Cameron 2017). One of the main purposes of migration is to minimize exposure to predation, especially during calving when young animals are particularly vulnerable (Fryxell and Sinclair 1988). Migratory barren-ground caribou tend to aggregate during calving and calve synchronously (most females giving birth within a week of each other). The hypothesized purpose of this phenomenon is to “swamp” or overwhelm the finite number of predators that exist within a relatively limited amount of space and time.
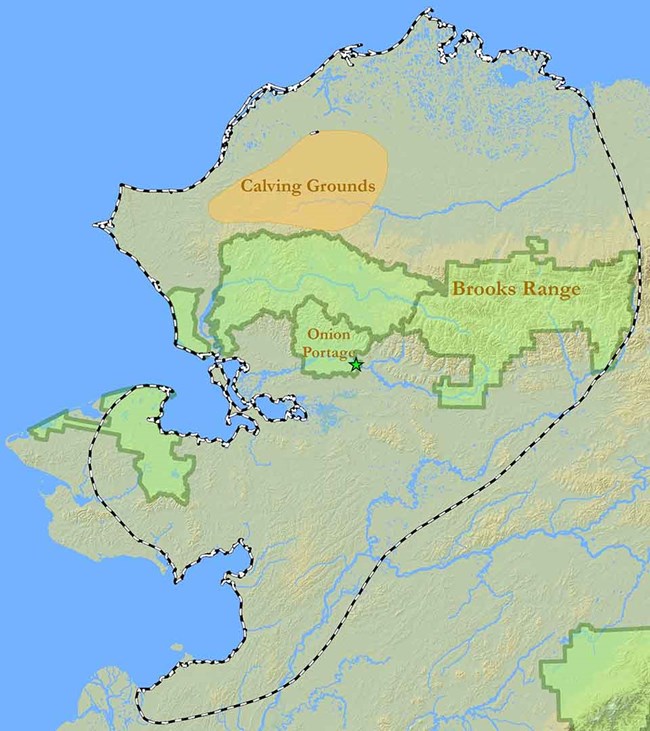
Lands managed by the National Park Service are in green. Onion Portage (green star), a traditional location to hunt caribou for thousands of years, lies within Kobuk Valley National Park.
For the Western Arctic Herd (WAH), which ranges over all of northwest Alaska, calving has taken place in the Utukok uplands, north of the Brooks Range, for at least a century (Figure 1; Lent 1966, Joly et al. 2011, Burch 2012). Another critical purpose of migration is to track the availability of abundant high-quality forage (Fryxell and Sinclair 1988). Green vegetation emerges this far north (69oN latitude) soon after calving and provides nutritious forage high in protein. Caribou need this high protein intake to fill nutritional deficits accumulated over the winter, produce milk, and gain body mass during the short summer months. Summer months are not easy on caribou; swarms of mosquitoes, biting flies, and other insect parasites harass caribou incessantly and are the primary driver of caribou movements during this time. Movement rates are greatest during this time of the year as caribou seek out cool and windy places for relief. It is at this time of year that the spectacular congregations of caribou occur, with groups numbering in the tens or even hundreds of thousands of individuals.

NPS/Kyle Joly
After insect harassment subsides, caribou disperse and can be found throughout the Brooks Range and the North Slope of Alaska. With the arrival of fall, most, but not all, caribou start to migrate south through the mountains. Commonly, caribou still cross the Kobuk River at Onion Portage, within Kobuk Valley National Park, during fall migration as they have done for ten thousand years (Anderson 1968, Joly and Cameron 2017). As forage quality decreases in vascular plants with the onset of fall, caribou begin to rely more heavily on lichens. Lichens dominate the winter diets of caribou and are much more abundant south of the Brooks Range (Joly et al. 2015), which may be an important factor driving the fall migration. Mid-winter is the time of energy conservation for caribou and is when movement rates are lowest. Depending on weather and snow conditions, caribou begin their northward spring migration in early April (Joly and Cameron 2017), back toward the calving grounds.

NPS/Kyle Joly
Caribou distribution, movements, and migratory patterns are known to be related to herd size (Messier et al. 1998, Ferguson and Messier 2000). Overgrazing of winter range may be a factor in this relationship. For example, as northwest Alaska elder Nelson Greist observes “if caribou numbers are too high, they over eat” and “when caribou over eat, they need to change” where they go. The size of the WAH naturally oscillates at the decadal scale (Joly et al. 2011). The herd hit a low of 75,000 animals in 1976, but quickly rebounded, reaching nearly 500,000 by 2003 (ADFG 2011). From 2003, the herd steadily declined to 201,000 caribou in 2016 (ADFG 2016), but again increased to 259,000 caribou in 2017 (ADFG 2018). As herd size increases, there is a tendency for its range to expand; as herd size decreases, its range often contracts. The combination of decreasing abundance and diminishing range size can produce extreme hardships for rural subsistence users that rely on caribou, particularly those at the edge of the herd’s range. As the WAH has declined, areas that often saw caribou in the past, such as Unalakleet, have not seen them for 15 years. As lichen abundance has declined on the eastern side of the Seward Peninsula, caribou have migrated out to the western Seward Peninsula to overwinter (Joly et al. 2007, Joly and Cameron 2017). Even annual shifts in migratory patterns can have profound impacts on communities in northwest Alaska that can experience large year-to-year swings in caribou availability even when the overall herd size is steady or growing.
On a global scale, long-distance, terrestrial migrations by large mammals are an imperiled phenomenon (Berger 2004). Encroachment of humans on the vast ranges used by migratory animals is one of the primary reasons for their endangerment. There has been relatively little development of northwest Alaska and thus caribou migrations continue to occur relatively unimpeded. A 50-mile- (80-km) long industrial road connecting a mine to its port site does intersect the western-most fall migration corridor of the WAH. The migration of some caribou traveling this route has been delayed by an average of 30 days (Wilson et al. 2016). Vehicular traffic stirs up dust along the road, which impacts vegetation and may alter caribou movements (Hasselbach et al. 2004, Chen et al. 2017). Other proposed development projects, like the 200-mile- (320-km) long road to the Ambler Mining District, could further constrain caribou movements in the area. Roads can have numerous impacts in addition to altering migratory movement, such as increasing vulnerability to vehicle collisions, predation, and hunting. Typically, it is not a single road or development that jeopardizes long-distance migrations, but the cumulative effects of many such projects. While caribou avoid dense vegetation, rough terrain, and wide sections of major rivers during their fall migration southward, current levels of sport hunting activity by people from outside the region does not appear to hinder migration (Fullman et al. 2017). However, many rural residents, such as Noatak elder Eugene Monroe, suggest that sport hunters that arrive by aircraft “disrupt the migration of the caribou” (Betcher 2016). Weather, climate change, and predators are other factors that may impact caribou movements. For example, hunter Lee Ballot, Sr. notes that caribou do not migrate when “it’s just too warm, that’s what triggers them to move, it is the cold” (Betcher 2016). Much additional research is required, but local knowledge can guide and enhance western-based scientific inquiries.
Large migratory caribou herds, such as the WAH, require vast spaces. While caribou are tolerant of some levels of development, vigilance, caution, and conservation are required to help maintain one of the greatest migratory spectacles in the world; if not just for its own existence, then for the people of northwest Alaska who are inextricably tied to caribou.
REFERENCES
Alaska Department of Fish and Game (ADFG). 2018.
Western Arctic Caribou Herd Increases After Years of
Decline. January 10, 2018 Press Release. Juneau, AK.
Alaska Department of Fish and Game (ADFG). 2016.
Update: July survey suggests Western Arctic Caribou Herd decline is leveling out. August 29, 2016 Press Release. Juneau, AK.
Alaska Department of Fish and Game (ADFG). 2011.
Caribou management report of survey-inventory activities 1 July 2008-30 June 2010. P. Harper (ed.). Alaska Department of Fish and Game, Juneau, Alaska. 345 pp.
Anderson, D. D. 1968.
A Stone Age campsite at the gateway to America. Scientific American 218: 24-33.
Banfield, A. W. F. 1962.
A revision of the reindeer and caribou, genus Rangifer. Natural Museum of Canada Bulletin 177. 137 pp.
Berger, J. 2004.
The last mile: how to sustain long-distance migration in mammals. Conservation Biology 18: 320-331.
Betcher, S. 2016.
Counting on caribou: Inupiaq way of life in northwest Alaska. Farthest North Films.
View Video
Burch, E. S., Jr. 2012.
Caribou Herds of Northwest Alaska 1850-2000. University of Alaska Press, Fairbanks.
Burch, E. S., Jr. 1972.
The Caribou/Wild Reindeer as a Human Resource. American Antiquity 37(3): 339-368.
Chen, W., S. G. LeBlanc, , H. P. White, C. Prevost, B. Milakovic, C. Rock, G. Sharam, H. O’Keefe, , L. Corey, B. Croft, A. Gunn, S. van der Wielen, A. Football, B. Tracz, J. S. Pellissey, and J. Boulanger. 2017.
Does dust from arctic mines affect caribou forage? Journal of Environmental Protection 8: 258-276.
Ferguson, M. A. D. and F. Messier. 2000.
Mass emigration of arctic tundra caribou from traditional winter range: population dynamics and physical condition. Journal of Wildlife Management 64: 168-178.
Fryxell, J. M. and A. R. E. Sinclair. 1988.
Causes and consequences of migration by large herbivores. Trends in Ecology and Evolution 3: 237-241.
Fullman, T. J., K. Joly, and A. Ackerman. 2017.
Effects of environmental features and sport hunting on caribou migration in northwestern Alaska. Movement Ecology 5(4): 11 pp. doi:10.1186/s40462-017-0095-z.
Guthrie, R. D. and J. V. Matthews. 1971.
The Cape Deceit fauna – Early Pleistocene mammalian assemblage from the Alaskan Arctic. Quaternary Research 1: 474-510.
Hasselbach, L., J. M. Ver Hoef, J. Ford, P. Neitlich, E. Crecelius, S. Berryman, B. Wolk, and T. Bohle. 2004.
Spatial patterns of cadmium and lead deposition on and adjacent to National Park Service lands near Red Dog Mine, Alaska: NPS Final Report. National Park Service Technical Report NRTR-2004-45. 59 pp.
Joly, K. and M. D. Cameron. 2017.
Caribou vital sign annual report for the Arctic Network Inventory and Monitoring Program: September 2015-August 2016. Natural Resource Report NPS/ARCN/NRR—2017/1398. National Park Service, Fort Collins, Colorado. 25 pp.
Joly, K., S. K. Wasser, and R. Booth. 2015.
Non-invasive assessment of the interrelationships of diet, pregnancy rate, group composition, and physiological and nutritional stress of barren-ground caribou in late winter. PLoS One 10(6): e0127586. doi:10.1371/journalpone.0127586.
Joly, K., D. R. Klein, D. L. Verbyla, T. S. Rupp, and F. S. Chapin, III. 2011.
Linkages between large-scale climate patterns and the dynamics of Alaska caribou populations. Ecography 34: 345-352.
Joly, K., R. R. Jandt, C. R. Meyers and M. J. Cole. 2007.
Changes in vegetative cover on Western Arctic Herd winter range from 1981-2005: potential effects of grazing and climate change. Rangifer Special Issue 17: 199-207.
Lent, P. C. 1966.
Calving and related social behavior in the barren-ground caribou. Zeitschrift fur Tierpsychologie 23: 701-756.
Messier, F., J. Huot, D. le Henaff, and S. Luttich. 1988.
Demography of the George River Caribou Herd: Evidence of population regulation by forage exploitation and range expansion. Arctic 41: 279-287.
Osborn, H. F. 1910.
The age of mammals in Europe, Asia and North America. The McMillan Company, New York. 676 pp.
Wilson, R. R., L. S. Parrett, K. Joly, and J. R. Dau. 2016.
Effects of roads on individual caribou movements during migration. Biological Conservation 195: 2-8.
Last updated: June 12, 2018
