Part of a series of articles titled Alaska Park Science - Volume 17, Issue 1. Migration: On the Move in Alaska.
Article
Connecting Taiga to Tropics: Swainson's Thrush as a Model for Nearctic-Neotropical Migration in Alaska

The Swainson’s Thrush (Catharus ustulatus) is a medium-sized songbird renowned for a beautiful spiraling flutelike song and an affinity for mature dense forests. Twice a year, the Swainson’s Thrush connects the boreal forests of North America to the tropical forests of Central and South America through its flight across the Western Hemisphere. About 200 bird species, primarily songbirds such as thrushes and warblers, are considered Nearctic-Neotropical migrants that fly thousands of miles annually between breeding and wintering areas. But why would an animal undertake such a perilous and energetically costly journey? Ultimately, long-distance migration exists because it increases a bird’s fitness which is the ability to raise young over its lifetime (Alerstam et al. 2003).
Alaska’s unique position on the globe, far north at the extremities of North America and the Pacific Ocean, and encompassing much of historical Beringia, provides the state with a fantastic diversity of birds. These birds must incorporate a wide variety of strategies to survive and successfully breed at latitudes greater than 60°N (Kessel and Gibson 1978). Most of Alaska’s 300 annually occurring bird species (Gibson et al. 2017) leave the state in the fall to overwinter further south. Where they go and how they get there evolved over time with changing climate and geography. Northern bird populations were thought to be particularly influenced by glacial cycling during the Pleistocene that shaped migratory pathways and created divergence in species (Avise and Walker 1998, Lovette 2005). In Alaska, we can trace the migratory routes of many songbirds to the recolonization of expanding northern forests by birds with the retreat of ice sheets (Pielou 1991).
The Swainson’s Thrush is an excellent model to illustrate post-glacial colonization of Alaska by migratory birds wintering in the New World tropics. Recent research has begun to paint a more complete story of the Swainson’s Thrush’s annual trip across continents. While observations and recovery of banded birds previously sketched an outline of the distribution of the two subspecies of Swainson’s Thrush throughout the year, application of new genetic, isotopic, and tracking methodologies across a large part of its breeding range has made this songbird’s migration one of the better understood in North America.
The two subspecies of Swainson’s Thrush, the “russet-backed” group (C. u. ustulatus) that breeds along the Pacific Coast of North America and the “olive-backed” inland group (C. u. swainsoni) that breeds in boreal forests across Canada and the United States, are distinguished by their plumage characteristics as well as by differences in migration routes, wintering areas, breeding habitat, and vocalizations (Mack and Yong 2000). Notably, banding data and observations during migration suggested that inland populations of Swainson’s Thrush made a long and circuitous flight east across North America before heading south, unlike their coastal cousins that take a direct route south to their wintering grounds in Central America (Brewer et al. 2000).
Genetic researchers began to investigate the divergence of the two subspecies of Swainson’s Thrush in depth in the early 2000s (Ruegg and Smith 2002, Reugg et al. 2006, Ruegg 2007). They found that the migratory pathway of the inland swainsoni group mirrors the post-glacial expansion of boreal forests and that subspecies likely diverged when ice sheets isolated populations during the last glacial maximum (Ruegg 2007). Where the two subspecies meet along the crest of the Cascade and Sierra Nevada Mountain Ranges, researchers found hybrid individuals that show physical and genetic traits that transition from usulatus to swainsoni along a gradient between coastal and interior ecosystems (Ruegg 2007). This research provided compelling evidence for the evolution of migratory pathways and the development of subspecies in songbirds through glacial cycling at northern latitudes; however, the complete story of the Swainson’s Thrush’s annual cycle were not known until tracking devices small enough to be deployed on songbirds were developed in the last decade.
Light-level geolocators have revolutionized research on small migratory birds (McKinnon et al. 2013). Previous tracking technology such as satellite and GPS transmitters were too heavy to deploy on smaller birds like thrushes, so the exact routes and wintering areas of specific breeding populations have been unknown. At less than a gram, geolocators are archival light-recording devices that record light levels in relation to time allowing researchers to calculate latitude and longitude based on day length and sun elevation angle (Hill 1994). Geolocators must be recovered to download the data, so they rely on the ability to recapture birds in subsequent years and therefore, sample sizes are generally low. Combined with genetic and isotope data, these units provide powerful empirical support for defining annual movements at a population level.
Researchers have deployed geolocators on Swainson’s Thrush at a number of sites across their western range including Point Reyes National Seashore in California, coastal and inland sites in British Columbia, Rocky Mountain National Park in Colorado, and most recently Denali National Park and Preserve in Alaska. The resulting location data confirmed the direct and cross-continent migration paths of the coastal and inland populations as well as the intermediate routes of hybrid and rocky mountain populations (Delmore et al. 2012, Cormier et al. 2013, Delmore and Irwin 2014, J. Beason [Bird Conservancy of the Rockies] unpublished data, NPS unpublished data). In addition to better outlining migratory pathways, geolocator data also helped define wintering areas for the various populations. Wintering areas ranged from western Mexico to central South America and showed strong migratory connectivity, or links between breeding and wintering locations, for populations in California, Coastal British Columbia, and Alaska (Figure 1, Comier et al. 2013, NPS unpublished data).
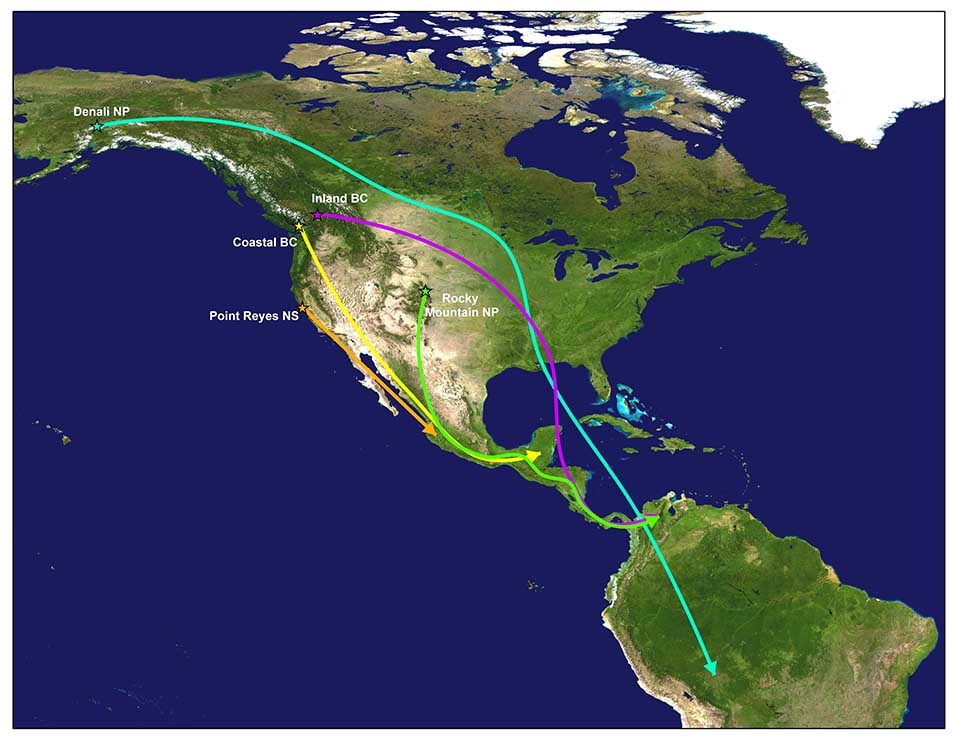
The resulting location data confirmed the direct and cross continent migration paths of the coastal and inland populations and also helped define wintering areas for the various populations (Delmore et al. 2012, Cormier et al. 2013, Delmore & Irwin 2014).
The ability to define specific wintering areas for breeding songbird populations has huge implications for conservation and protection since these birds spend more of each year in the tropics than in their northern nesting grounds. While widespread declines in Nearctic-Neotropical migratory bird populations have been reported in the literature since the 1980s (Rappole and McDonald 1994), current research has highlighted how complex the population dynamics of migratory birds may be as populations respond temporally and spatially to various factors at breeding and wintering areas as well as during migration (Faaborg et al. 2010). Managers of migratory birds have to know what habitats these wide-ranging birds are using throughout the year to really understand what factors might be driving population declines. For the Swainson’s Thrush, we now know that factors driving population trends in birds breeding in Rocky Mountain National Park will likely be very different than those affecting Denali National Park and Preserve’s thrushes since the areas used by each population overlap very little across the year. To protect Swainson’s Thrushes and other migratory birds, National Park Service managers will need to collaborate with national and international partners responsible for the conservation of specific habitats used by different breeding populations throughout their annual cycle. A greater understanding of where birds go when they leave park boundaries is critical to their conservation.
Acknowledgements
I’d like to acknowledge the other principal investigators leading the bird migration research in Denali National Park and Preserve: Carol McIntyre, Scott Weidensaul, and Iain Stenhouse. Emily Williams, Dave Merz, Jason Reppert, Jill Boeslma, George Gress and other field staff were critical to the success of the project in Denali. I’d also like to thank Jason Beason at the Bird Conservancy of the Rockies and Diana Humple at Point Blue for sharing their research results and field techniques. Funding for the Denali research is provided by the National Park Service with additional support from Alaska Geographic, the Denali Education Center, the Murie Science and Learning Center, and Camp Denali.
REFERENCES
Alerstam, T., A. Hedenström, and S. Åkesson. 2003.
Long-distance migration: evolution and determinants. Oikos 103: 247-260.
Avise, J. C. and D. E. Walker. 1998.
Pleistocene phylogeographic effects on avian populations and the speciation process. Proceedings of the Royal Society of London B: Biological Sciences 265: 457-463.
Brewer, D., A. Diamond, E. J. Woodsworth, B. T. Collins, and E. H. Dunn. 2000.
Canadian atlas of bird banding. Puslinch, Ontario: Canadian Wildlife Service.
Cormier, R. L., D. L. Humple, T. Gardali, and N. E. Seavy. 2013.
Light-level geolocators reveal strong migratory connectivity and within-winter movements for a coastal California Swainson’s Thrush (Catharus ustulatus) population. The Auk 130: 283-290.
Delmore, K. E. and D. E. Irwin. 2014.
Hybrid songbirds employ intermediate routes in a migratory divide. Ecology Letters 17: 1211-1218.
Delmore, K. E., J. W. Fox, and D. E. Irwin. 2012.
Dramatic intraspecific differences in migratory routes, stopover sites and wintering areas, revealed using light-level geolocators. Proceedings of the Royal Society of London B: Biological Sciences 279: 4582-4589.
Gibson, D. D., L. H. DeCicco, R. E. Gill Jr., S. C. Heinl, A. J. Lang, T. G. Tobish Jr., and J. J. Withrow. 2017.
Checklist of Alaska Birds, 23rd Edition – 2017. University of Alaska Museum, Fairbanks 99775-6960.
Faaborg , J., R. T. Holmes, A. D. Anders, K. L. Bildstein, K. M. Dugger, S. A. Gauthreaux, P. Heglund, K. A. Hobson, A. E. Jahn, D. H. Johnson, S. C. Latta, D. J. Levey, P. P. Marra, C. L. Merkord, E. Nol, S. I. Rothstein, T. W. Sherry, T. S. Sillett, F. R. Thompson III, and N. Warnock. 2010.
Conserving migratory land birds in the New World: Do we know enough? Ecological Applications 20: 398-418.
Hill, R. D. 1994.
Theory of geolocation by light levels. Elephant seals: population ecology, behavior, and physiology. University of California Press, Berkeley, pp.227-236.
Kessel, B. and D. D. Gibson. 1978.
Status and distribution of Alaska birds (No. 1). Cooper Ornithological Society.
Lovette, I. J. 2005.
Glacial cycles and the tempo of avian speciation. Trends in Ecology & Evolution 20: 57-59.
Mack, D. E. and W. Yong. 2000.
Swainson’s Thrush (Catharus ustulatus). The birds of North America 540: 32.
McKinnon, E. A., K. C. Fraser, and B. J. Stutchbury. 2013.
New discoveries in landbird migration using geolocators, and a flight plan for the future. The Auk 130: 211-222.
Pielou, E. C. 1991.
After the Ice Age: the return of life to glaciated North America. The University of Chicago Press.
Rappole, J. H. and M. V. McDonald. 1994.
Cause and effect in population declines of migratory birds. The Auk 1: 652-660.
Ruegg, K. 2007.
Divergence between subspecies groups of Swainson’s Thrush (Catharus ustulatus ustulatus and C. u. swainsoni). Ornithological Monographs 63: 67-77.
Ruegg, K. C., R. J. Hijmans, and C. Moritz. 2006.
Climate change and the origin of migratory pathways in the Swainson’s thrush, Catharus ustulatus. Journal of Biogeography 33: 1172-1182.
Ruegg, K. C. and T. B. Smith. 2002.
Not as the crow flies: a historical explanation for circuitous migration in Swainson’s thrush (Catharus ustulatus). Proceedings of the Royal Society of London B: Biological Sciences 269: 1375-1381.
Last updated: December 30, 2019
