
Michael Klensch

D. Lummerzheim
Abstract
The aurora has fascinated observers at high latitudes for centuries, but only recently have we begun to understand the processes that cause it. This article discusses the mechanisms that are responsible for the colors of the aurora. Observations of color balance in aurora can provide us with information about the physical processes in the near Earth space that cause aurora. High-resolution spectral observations let us understand how the upper atmosphere is affected by aurora
A Brief History of Understanding the Aurora
Descriptions of aurora, or the northern lights, go as far back as written history. 2,300 years ago, Aristotle saw curtains of light in the sky and called the phenomena “chasmata” to indicate that the cause was cracks in the sky, allowing in light from beyond the heavenly sphere. In his book Majestic Lights, Eather (1980) presents several quotes from the Bible that most likely refer to aurora.
The first recorded use of the words “northern lights” to describe the aurora was in 1230, in a book titled The King’s Mirror. The author wrote the book to prepare Norwegian King Magnus Lagabøte for his duties as a ruler.

Poul Jensen
The term aurora borealis originated in the 1600s, when Galileo combined the word “aurora,” the Latin word for “dawn,” with the term “boreal,” the Greek word for “north.” Aurora also appears in the southern hemisphere, where it is called the “aurora australis.” Since there is very little populated landmass at high southern latitudes, there are no known historical and mythological references to the southern aurora. Although the native people from New Zealand must have seen aurora on occasion, Captain Cook is considered the discoverer of the aurora australis; he saw it in 1773 on his voyage around the southern tip of South America.
At mid-latitudes, people rarely see the northern lights. Aurora is visible at mid-latitudes during the largest magnetic storms, but it is dominated by red colors. In ancient times when the aurora appeared overhead, people often associated the aurora with good or bad omens and sometimes considered it a manifestation of activities of heavenly spirits or gods. The peoples who lived at high latitude and who had a regular display of the aurora held similar beliefs.

Carl Johnson
In the Middle Ages, scientists came up with other guesses as to what was behind the northern lights: they suggested that the light of the aurora was sunlight reflected by ice crystals in the air, the glow of glacier ice near the pole, or a light emanating from the edge of Earth. In the eighteenth century, scientists discovered a connection between the aurora and disturbances in Earth’s magnetic field and associated aurora with sunspots. But it took until the end of the twentieth century before a satisfactory explanation of the aurora, its colors, and the mechanisms behind it emerged.

Michael Klensch
The Processes that Cause Aurora
The light of the aurora is generated by atoms and molecules of the air when they are struck with energetic particles from space. These energetic particles come from the volume of space just above the aurora, and are accelerated by plasma physics processes that are still under investigation. But we do have a fairly good understanding of the general processes and the flow of energy that feeds these processes. We can model the aurora and are now gaining the understanding to forecast its appearance (Lummerzheim 2007).
Charged particles, like those that cause the aurora, can generally only travel along the direction of the magnetic field. This shapes the aurora into curtain and ray-like structures (Figure 2). Following the magnetic field up from the aurora, we get to the auroral acceleration region, about 620-6,200 miles (1,000-10,000 km) above the earth. The smaller scale structures like rays, small curls, and thin curtains shown in Figure 3 represent structures in the acceleration processes. This region is connected to the outer magnetosphere by electric currents. Large-scale structures, like multiple parallel arcs (Figures 4-5) and spirals that fill almost the entire sky (Figure 6) show the spatial pattern of these currents. The magneto-sphere is the region of space around Earth that is controlled by Earth’s magnetic field. Its diameter is about 30 Earth radii, and outside of the magnetosphere is the solar wind. The magnetosphere forms an obstacle for the solar wind, which has to flow around it. This interaction of the solar wind with the magnetosphere provides the energy that eventually accelerates the auroral electrons in the inner magnetosphere. Strong solar activity causes strong variations in the solar wind; byproducts of this space weather are therefore geomagnetic storms and aurora.

Poul Jensen
Light Emission in Aurora
When energetic electrons strike an atom or molecule, they slow down and transfer some of their energy to that atom or molecule. The molecules can store this energy only for a very short time, and then radiate the energy away as light. Some molecules get dissociated into atoms in this process, and some molecules and atoms get ionized. At the altitude where aurora occurs, above about 62 miles (100 km), the air is thin enough that oxygen can exist in atomic form, while the air that we breathe contains only molecular oxygen. During the day, the ultraviolet sunlight splits the molecular oxygen into atoms, while at night the aurora continues this process.
When an atom or molecule emits light as a photon, to rid itself of its excess energy, that photon has a wavelength that is characteristic for that atom. We perceive wavelength as color. Laboratory experiments can reproduce these light-emitting processes by forcing a current through an evacuated glass tube that contains a small amount of a selected gas. The study of these light-emitting processes led to the understanding of atoms early in the twentieth century, and to the discovery of quantum mechanics. Because each type of atom or molecule emits colors unique to it, we can use the colors of the aurora to determine the atmospheric composition at the auroral altitude.

Poul Jensen
The time that a molecule or atom can store the energy that it gained in a collision is very short, typically between 1/1000 and less than 1/1,000,000 of a second. Atomic oxygen is one notable exception, and the excited state that causes the most common auroral emission, the green line, has a lifetime of 0.7 seconds. When an excited atom takes that long to radiate away the internally stored energy, other processes, chemical reactions or collisions, compete with the radiation process for that energy. The denser the air is, the more frequent are the collisions between the atoms and molecules. Below the altitude of about 59 miles (95 km), collisions are so frequent that the green oxygen line has no chance to be emitted. All the energy that is put into the oxygen atom is lost before the 0.7-second lifetime allows radiation. This determines the bottom edge of the green emission in aurora.

Poul Jensen
However, the auroral electrons sometimes have enough energy to give them the punch to penetrate deeper than that into the atmosphere. When that happens, only emissions with a much shorter lifetime are possible. The most abundant gas is molecular nitrogen, and it radiates promptly in deep blue and red colors. Mixing these together gives purple. The bottom edge of a green auroral curtain gets this purple color when auroral elec-trons are accelerated to very high energy (Figures 7-8).
On occasion the aurora gets a deep red color. This comes from higher altitudes, around 120-180 miles (200-300 km). It is again the oxygen atom that is responsible for this color. The oxygen atom has an excited state for this red line emission with a mean lifetime of 100 seconds, and only at very high altitudes are collisions infrequent enough to allow this radiation to be emitted (Figure 9). Since the long life-time of the oxygen red line also allows the aurora to move before it radiates, the de-tailed structure in auroral curtains is also washed out in these emissions (Figure 10).

Michael Klensch
Relating the Color to Physical Processes
The energy of auroral electrons determines how deep into the atmosphere these particles penetrate. Since auroral emissions are characteristic of the altitude where they originate, we can use the color balance of the aurora to determine the energy of the auroral electrons. The auroral brightness depends on the rate of incoming auroral electrons. In analogy to electric currents we can say that the color tells us the voltage, the brightness tells us the current.
During very large magnetic storms the aurora is visible from mid and low latitudes. It is common to have very bright red auroral displays during such large storms. Notable were the magnetic storms on Halloween and late November in 2003, when red aurora was seen above the Mediterranean, Florida, and the entire U.S. The outstanding brightness and dominance of high altitude red oxygen emissions indicate that during such storms the magnetosphere has very large currents flowing, while the auroral acceleration only produces low energy electrons.

Michael Klensch
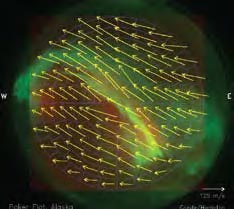
In addition to looking at the color balance and brightness, we can measure the wavelength of individual emission lines in the aurora with very high accuracy. This allows us to determine the Doppler shift of emission lines. The Doppler effect for light emission causes a shortening of the wavelength of the emission if the emitting atom or molecule is moving toward the observer, and a lengthening of the wave-length if it is moving away. A shorter wave-length means a color closer to the blue end of the spectrum; longer wavelength means a shift to red. In aurora, these shifts are miniscule, but can be observed with high spectral resolution instruments, in particular Fabry-Perot interferometers (FPI). Because the red and green line emissions from atomic oxygen are so long lived, they are good candidates for FPI observations. The long lifetime ensures that the atoms are drifting with the wind, and their velocity is not affected by the collision with the auroral electron that caused the excitation in the first place. The small shift in wavelength can thus be used to measure the ambient wind at the altitude of the aurora.
Michael Klensch
These FPI wind observations only give the component of the wind velocity along the line of sight, the component toward or away from the observing station. By using model constraints or by placing three such instruments in separate loca-tions we can reconstruct the actual wind vector by measuring three components of it. Figure 11 shows a composite of the auroral brightness with the deduced wind vectors at 150 miles (240 km) altitude superimposed. This example (Conde et al. 2008) shows that the wind is affected by the aurora as the direction and speed changes right at the position of the auroral curtain.
The top end of the auroral curtains and rays sometimes show a deep blue color. This is indicative of still another light emission process. The auroral electrons not only produce light emitting excited atoms and molecules, they also ionize some molecules. These ions can then be pulled upward by electric fields in the aurora and reach altitudes high enough that under some conditions they will be exposed to sunlight. This sunlight then scatters off these ions. There exists a blue emission of the molecular nitrogen ion that is particularly strong in scattering sunlight, which is why we see a blue upper end of the auroral curtains (Figure 13).

Michael Klensch
Putting it all Together
The observations of the colors of the aurora, either in a broader sense by looking at the overall color balance, or by detailed spectroscopic methods, can teach us much about the physical processes that cause aurora and the effects that aurora has on the upper atmosphere. The color balance tells us the altitude of aurora. We can relate that to the processes that accelerate auroral electrons in the near earth space, and we can see the evolution of electric currents that flow in the magnetosphere. High-resolution spectroscopy lets us see the wind in the upper atmosphere and how it is changed by the aurora. High altitude blue aurora tells us that ions that are generated in the aurora are pulled out of the atmosphere into space. And the colors themselves tell us the composition of the atmospheric gas at the altitude of the aurora.

Jan Curtis
Acknowledgements
I would like to thank Mark Conde for suggestions and comments, and the people who have provided the photographs of the aurora for this article: Poul Jensen at the Geophysical Institute at the University of Alaska Fairbanks; Michael Klensch from Skagway, Alaska; Carl Johnson from Anchorage, Alaska; and Jan Curtis from Oregon. More aurora photos by these photographers can be found at their websites:
www.gfy.ku.dk/~flyvholm (P. Jensen)
www.muk.uni-hannover.de/~theusner/polarlicht/ (M. Theusner)
www.alpenglowphoto.net (M. Klensch)
climate.gi.alaska.edu/Curtis/aurora/ aurora.html (J. Curtis)
www.carljohnsonphoto.com (C. Johnson)
References
Conde, M.G., C. Anderson, and C. Ander-son. 2008.
New results from the Poker Flat all-sky imaging Fabry-Perot spectrometer. CEDAR workshop, Zermatt, UT.
Eather, R.H. 1980.
Majestic Lights: The Aurora in Science, History, and the Arts. American Geo-physical Union. Washington DC.
Lummerzheim, D. 2007.
Modeling and forecasting aurora. Computing in Science and Engineering 9(5):53-61.
Part of a series of articles titled Alaska Park Science- Volume 8 Issue 1: Connections to Natural and Cultural Resource Studies in Alaska's National Parks.
Last updated: October 26, 2021
